Autonomous Pharmaceutical CSTR Process Intelligence Team
Introduction
Pharmaceutical manufacturing excellence depends on maintaining precise control of Continuous Stirred Tank Reactor (CSTR) conditions while balancing competing objectives of product quality, process efficiency, equipment reliability, and regulatory compliance. Yet most pharmaceutical CSTR operations struggle with traditional control approaches that operate in isolation, missing critical interactions between process variables, equipment health, quality parameters, and compliance requirements that directly impact both product quality and regulatory compliance.
The Pharmaceutical CSTR Process Intelligence Team represents an advanced approach to pharmaceutical process control - specialized AI agents that work together to monitor, control, and optimize key aspects of continuous stirred-tank reactor operations. Unlike traditional single-loop controllers that address isolated parameters, this collaborative approach enables understanding of how process variables, equipment health, quality requirements, regulatory compliance, and operational efficiency interact in complex pharmaceutical manufacturing environments.
The Multi-Dimensional Pharmaceutical CSTR Control Challenge
Achieving optimal pharmaceutical CSTR performance requires coordination across multiple interdependent process variables while maintaining strict regulatory compliance and quality standards. Traditional control approaches often treat each parameter independently, missing important interactions that affect overall reactor performance, product quality, and compliance status.
Key Challenge Areas
Process Control Complexity
- Temperature control affects reaction rates, product quality, and process validation simultaneously
- Concentration variations cascade through the system, impacting downstream processes and final product specifications
- Pressure fluctuations can trigger safety shutdowns or compromise manufacturing conditions
- Flow rate disturbances require coordinated adjustments across multiple control loops
- Traditional controllers may oscillate when strong process interactions occur
Equipment Reliability and Maintenance
- Equipment failures can develop without warning, causing costly production disruptions
- Heat exchanger efficiency degradation impacts energy consumption and temperature control
- Mechanical component deterioration creates contamination risks and maintenance requirements
- Vibration monitoring indicates potential equipment problems that require attention
- Maintenance scheduling often conflicts with production demands
Quality and Compliance Integration
- Process variability can compromise product quality and batch consistency
- pH variations affect product stability and require monitoring
- Regulatory compliance requires continuous data integrity monitoring
- Electronic batch record documentation demands real-time parameter tracking
- Process validation protocols require statistical process control
Resource Utilization and Planning
- Equipment utilization can be suboptimal due to inefficient scheduling and changeovers
- Production planning may lack integration with equipment health and maintenance requirements
- Inventory levels may exceed optimal targets due to coordination challenges
- Energy consumption may run above optimal levels
- Schedule adherence problems can create downstream production impacts
Compound Impact Statement
These interconnected challenges create a complex optimization problem where traditional pharmaceutical manufacturing approaches face limitations: process excursions can compromise product quality and regulatory compliance, equipment issues disrupt schedules and increase costs, manual resource planning may lead to suboptimal utilization, and systems often operate below potential while maintaining compliance requirements. Addressing this complexity requires more than individual controls - it demands intelligent coordination across all aspects of pharmaceutical CSTR operation. The Pharmaceutical CSTR Process Intelligence Team takes a collaborative approach, with specialized agents working together to achieve comprehensive process optimization.
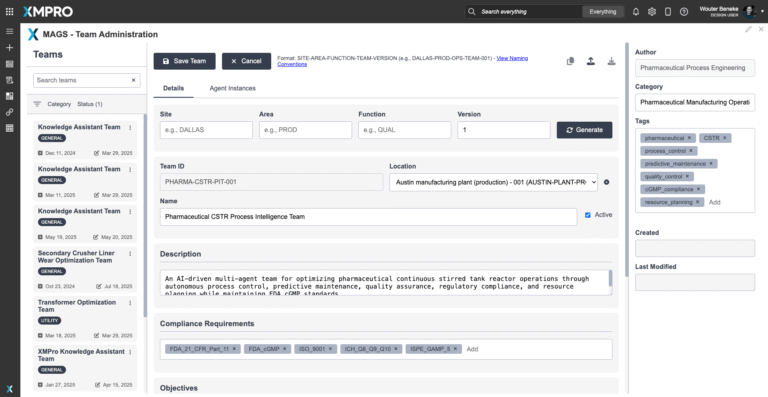
XMPro Autonomous Multi Agent Pharmaceutical CSTR Process Intelligence Team
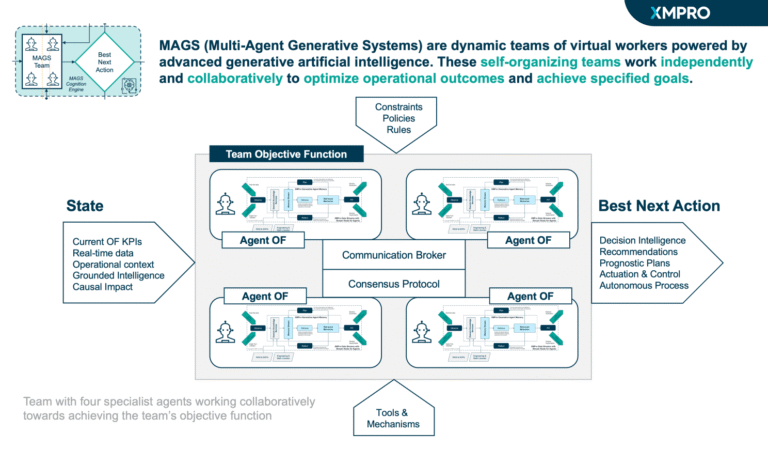
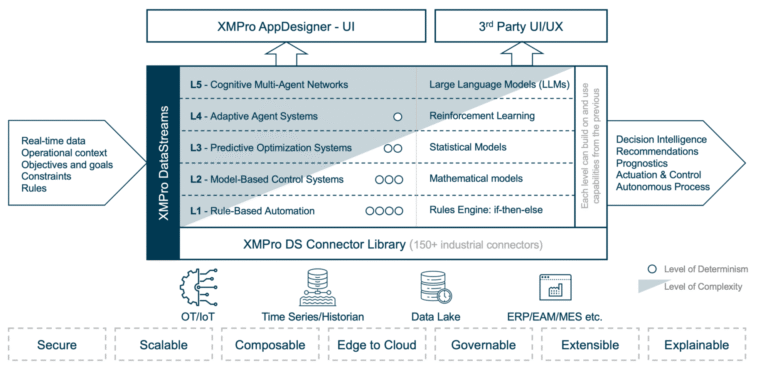
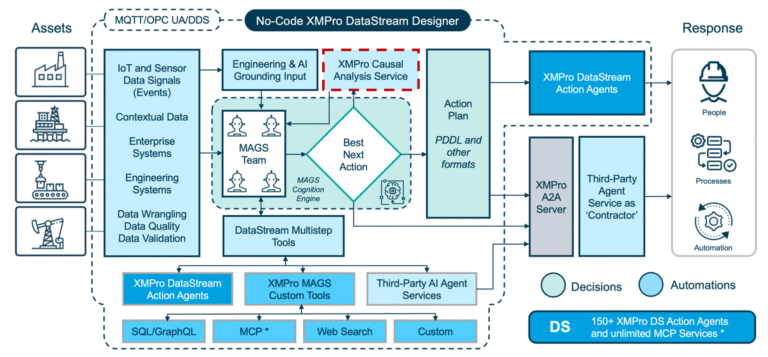
XMPro's Multi-Agent Generative Systems (MAGS) deploy autonomous AI teams that optimize pharmaceutical Continuous Stirred-Tank Reactor operations with regulatory compliance. Unlike traditional controllers or consumer AI, MAGS teams operate with bounded autonomy, using Composite AI grounded in chemical engineering principles, pharmaceutical process knowledge, and real-time process data. Every agent action is transparent and explainable, with reasoning paths that can be inspected. Decisions are grounded in process understanding and constrained by safety rules and regulatory requirements. XMPro's architecture enforces data validation, control boundaries, and compliance governance, making MAGS a secure framework for operational AI in pharmaceutical manufacturing environments.
Agents follow an observe, reflect, plan, and act cycle, supported by contextual reasoning and process memory. They maintain awareness of reactor conditions through continuous monitoring and collaborate to anticipate and address emerging issues through coordinated responses and compliance validation.
Teams dynamically adjust control strategies as process conditions evolve, balancing competing priorities in real time while maintaining audit trail integrity. Collective learning across agents ensures continuous improvement, with each agent's discoveries enhancing the team's overall effectiveness and regulatory compliance over time.
- Multi-Agent Collaboration: Specialized agents (Process Controller, Equipment Reliability, Quality Guardian, Compliance Monitor, Resource Planner) work together to optimize pharmaceutical CSTR performance
- Parametric Configuration: Customizable to your specific needs—from safety thresholds to optimization weights to control parameters—ensuring the system works your way
- Continuous Cognitive Cycle: Each agent follows observe-reflect-plan-act cycles, continuously learning and improving from process outcomes
- Real-Time Digital Twin: Digital representation of your pharmaceutical CSTR operations with simulation capabilities for process dynamics
- Composite AI Approach: Combines engineering models, statistical process control, and expert control strategies with compliance validation
- Graduated Autonomy: Start with monitoring and recommendations, progress to autonomous process optimization while maintaining regulatory oversight
- Regulatory Integration: Built-in compliance features with electronic batch records, audit trail management, and regulatory standards support
MAGS Team Composition
Meet Your Intelligent AI Team For Pharmaceutical CSTR Optimization
CSTR Process Optimization Agent
(Yield & Output Maximizer)
Optimizes fundamental CSTR process parameters for pharmaceutical manufacturing
Key Expertise: Temperature stability, flow rate control, pressure management, residence time optimization, energy efficiency
Team Contribution: Implements coordinated control actions based on inputs from all other agents, balancing process optimization with quality and compliance
CSTR Equipment Reliability Agent
(Mechanical Integrity Specialist)
Monitors equipment health and predicts maintenance needs
Key Expertise: Motor performance analysis, vibration monitoring, heat exchanger efficiency, mechanical seal integrity, predictive maintenance
Team Contribution: Provides equipment health insights and maintenance timing recommendations, ensuring equipment protection while maximizing availability
CSTR Quality Control Agent
(Process Consistency Guardian)
Manages batch-level quality and consistency assessment requirements
Key Expertise: Batch disposition, quality trending, regulatory reporting, deviation management, statistical process control
Team Leadership: Takes charge when quality risks emerge, coordinating team response to maintain pharmaceutical product specifications, batch consistency and compliance
CSTR Regulatory Compliance Agent
(Data Integrity & Documentation Specialist)
Ensures regulatory compliance and audit trail integrity
Key Expertise: Regulatory compliance tracking, documentation management, audit trail maintenance
Team Interaction: Validates all agent decisions against regulatory requirements and maintains complete documentation for audit readiness
CSTR Resource Planning & Scheduling Agent
(Production Planning Specialist)
Optimizes production planning and resource allocation
Key Expertise: Equipment utilization, batch sequencing, changeover optimization, inventory management, maintenance coordination
Team Interaction: Coordinates production planning with all operational constraints to maximize facility utilization
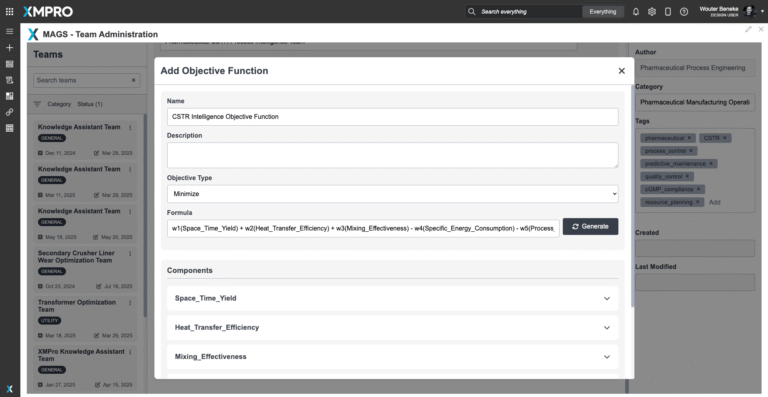
Team Objective Function
Collective Success Metrics - Objective Function
The Pharmaceutical CSTR Process Intelligence Team operates on a parametric mathematical framework that can be tuned to your specific pharmaceutical manufacturing objectives. Unlike rigid control systems, optimization parameters can be adjusted to match your production priorities and regulatory requirements.
Parametric Team-Level Formula:
Maximize CSTR_Performance = w₁(Process_Yield) + w₂(Heat_Transfer_Efficiency) + w₃(Mixing_Effectiveness) + w₄(Equipment_Utilization) + w₅(Quality_Consistency) - w₆(Energy_Consumption) - w₇(Process_Variability) - w₈(Compliance_Risk)Component Definitions (all with configurable parameters):
- Process_Yield: Product output relative to reactor capacity and time
- Heat_Transfer_Efficiency: Heat management effectiveness
- Mixing_Effectiveness: Mixing quality and uniformity
- Equipment_Utilization: Actual operating time relative to available time
- Quality_Consistency: Consistency of critical quality parameters
- Energy_Consumption: Total energy input per unit product output
- Process_Variability: Variation in critical process parameters
- Compliance_Risk: Regulatory compliance deviations and documentation gaps
Configurable Weighting Factors (example default values):
- Process Yield (w₁ = 0.20): Adjustable based on throughput priorities
- Heat Transfer Efficiency (w₂ = 0.15): Modifiable for energy-critical operations
- Mixing Effectiveness (w₃ = 0.15): Adjustable for homogeneity-critical products
- Equipment Utilization (w₄ = 0.20): Scalable based on capacity utilization priorities
- Quality Consistency (w₅ = 0.15): Adjustable for quality-critical pharmaceutical products
- Energy Consumption (w₆ = 0.05): Adjustable based on energy cost priorities
- Process Variability (w₇ = 0.05): Configured to ensure pharmaceutical consistency
- Compliance Risk (w₈ = 0.05): Configured to maintain regulatory compliance
Dynamic Parameter Adjustment Examples:
- High-Value Product Mode: Increased Quality and reduced Variability weights
- Energy Optimization Mode: Increased Energy and Heat Transfer efficiency weights
- Maximum Throughput Mode: Increased Yield and Utilization weights
- Equipment Protection Mode: Increased Utilization and Mixing effectiveness weights
Individual Agent Parameters (examples):
- Process Control Agent:
- Temperature control responsiveness: Configurable based on product thermal sensitivity
- Flow rate tolerance: Adjustable based on residence time requirements
- Control loop adaptation rate: Configurable based on process dynamics
- Equipment Reliability Agent:
- Vibration monitoring sensitivity: Adjustable based on equipment criticality
- Motor load protection thresholds: Configurable based on motor specifications
- Predictive maintenance confidence levels: Adjustable based on historical accuracy
Configurable Operational Constraints:
- Temperature limits: Adjustable based on pharmaceutical product requirements
- Pressure boundaries: Configurable based on vessel design specifications
- pH control ranges: Customizable based on product stability requirements
- Regulatory parameters: Maintained with configurable warning thresholds
- Custom pharmaceutical constraints: Configurable based on specific product and process requirements
Smart Parameter Management:
- Product Recipe Library: Save and switch between parameter sets for different pharmaceutical products
- A/B Testing: Run different parameters on parallel reactors or sequential batches with validation
- Auto-Tuning: Parameter optimization over time based on process outcomes and compliance
- Simulation Mode: Test parameter changes before deployment using digital twin models
Individual Agent Contributions:
- CSTR Process Control Agent optimizes Process_Yield and Heat_Transfer_Efficiency through temperature and flow control strategies
- CSTR Equipment Reliability Agent maximizes Equipment_Utilization while optimizing Energy_Consumption through predictive maintenance and equipment optimization
- CSTR Quality Control Agent ensures Quality_Consistency and minimizes Process_Variability through statistical process control
- CSTR Regulatory Compliance Agent minimizes Compliance_Risk through continuous data integrity monitoring and audit trail management
- CSTR Resource Planning & Scheduling Agent maximizes Equipment_Utilization and coordinates production planning to balance all objectives within regulatory constraints
Synergistic Effects
- Process control accuracy improves through multi-agent correlation analysis across temperature, mixing effectiveness, and equipment health
- Process interactions discovered through cross-functional data integration between quality parameters and process variables
- Resource optimization opportunities become apparent through collaborative planning and equipment health coordination
Risk Distribution
- No single point of failure - agents provide backup analysis capabilities for critical pharmaceutical process monitoring
- Multiple perspectives reduce blind spots in regulatory risk assessment and data integrity management
- Distributed decision-making prevents isolated optimization errors that could compromise product quality or compliance
- Redundant monitoring ensures critical parameter deviations and compliance issues are detected
Comprehensive Coverage
- Complete pharmaceutical process lifecycle management from batch startup through steady-state to completion and changeover
- Continuous monitoring without human fatigue or shift-change gaps in process surveillance and regulatory compliance
- Simultaneous optimization across all CSTR parameters with intelligent trade-off management and regulatory constraint adherence
- Proactive intervention before process upsets cascade across quality, compliance, equipment health, and resource utilization metrics
Adaptive Response
- Dynamic control strategy adjustment based on current pharmaceutical production priorities and regulatory requirements
- Intelligent trade-off management between competing objectives while maintaining compliance
- Real-time rebalancing of control focus as reaction conditions, equipment status, and regulatory priorities change
- Continuous refinement of process control thresholds and safety margins based on operational learning
Accelerated Learning
- Each agent's insights train the others, multiplying pharmaceutical process learning speed across all operational domains
- Process and equipment pattern recognition improves across all parameters simultaneously
- Best pharmaceutical manufacturing practices automatically propagate throughout the team for consistent performance
- Collective memory prevents repeated process control mistakes, equipment failures, and regulatory compliance issues
Team Dynamics Summary
- Real-time data sharing at appropriate intervals across all agents with critical parameter updates for safety-critical variables
- Priority alerts trigger immediate team-wide coordination for safety, quality, or regulatory compliance concerns
- Structured information exchange ensuring no critical pharmaceutical process insights are missed between control cycles
- Contextual updates that help each agent understand the broader pharmaceutical process state and regulatory compliance context
- Hierarchical with regulatory oversight - Regulatory Compliance Agent has validation authority over all actions affecting compliance
- Process Control Agent leads during normal pharmaceutical process control and optimization decisions
- Quality Control Agent leads for product quality-related decisions and process consistency requirements
- Equipment Reliability Agent influences process optimization based on equipment health and maintenance requirements
- Resource Planning Agent coordinates production scheduling and resource allocation with all operational constraints
- Regulatory Compliance Agent validates all decisions against regulatory requirements and maintains audit trail integrity
- Built-in priority matrix: Patient Safety > Regulatory Compliance > Equipment Protection > Process Quality > Resource Efficiency > Energy Optimization
- Automated trade-off analysis integrates insights from all agents when resolving conflicting pharmaceutical control actions
- Escalation to human operators for decisions exceeding confidence thresholds or requiring regulatory interpretation, supported by comprehensive team analysis
- Learning from resolved conflicts is shared across all agents to continuously improve future pharmaceutical process coordination
- Dynamic responsibility allocation based on current pharmaceutical process phase and regulatory requirements
- Agents increase monitoring intensity in their domain when pharmaceutical process conditions approach warning thresholds or regulatory limits
- Collaborative problem-solving with lead agent coordination based on the dominant pharmaceutical concern
- Automatic workload redistribution during high-stress periods, equipment emergencies, or regulatory compliance events
- Immediate human notification for patient safety risks, critical process deviations, or regulatory compliance issues, triggered by any agent
- Structured escalation based on pharmaceutical process impact severity, regulatory implications, and agent confidence levels
- Context-rich alerts with full team analysis including process history, regulatory status, equipment health, and recommended corrective actions
- Post-escalation learning shared across all agents to improve future autonomous handling of similar pharmaceutical process situations