Pharmaceutical Cold Chain Crisis Response Team
Introduction
Pharmaceutical cold chains don’t fail because data is missing — they fail when quality, supply, logistics, and compliance teams cannot respond quickly enough to protect product integrity. A temperature excursion, customs delay, or documentation gap can cascade into product loss, regulatory penalties, and risks to patient safety if not addressed in time. Manual processes and siloed tools slow responses and make coordination inconsistent.
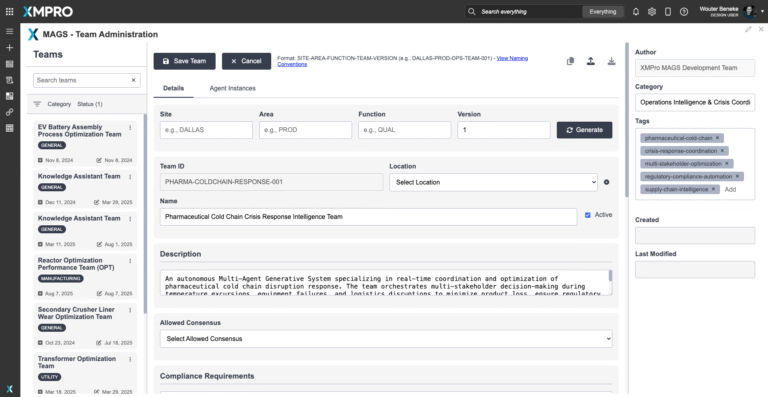
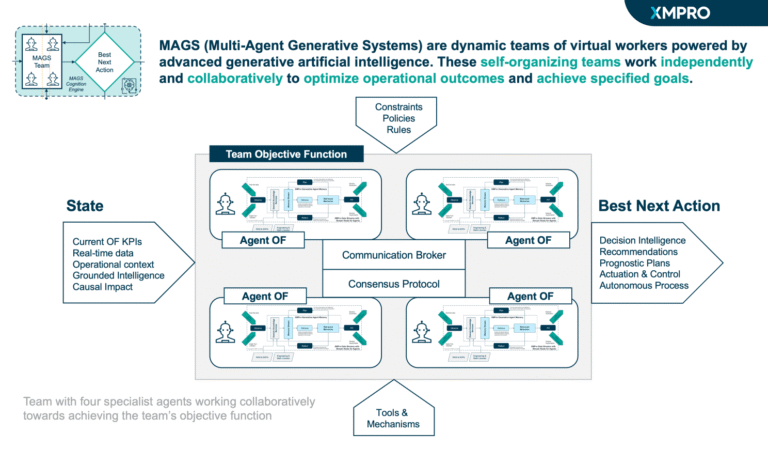
The Pharmaceutical Cold Chain Crisis Response MAGS Team solves this by combining five specialized agents — Quality Assurance, Supply Continuity, Logistics Optimization, Regulatory Compliance, and Crisis Detection & Escalation — into a governed multi-agent system. Each agent contributes its own domain expertise, reasoning cycles, and objective functions, while collaborating through XMPro’s MAGS consensus mechanism.
Unlike traditional monitoring tools, this team balances safety, compliance, efficiency, and continuity in real time. Every alert, recommendation, and escalation is transparent, explainable, and fully auditable. The result is a cold chain that responds as a connected, governed system — protecting patients, preserving product, and building trust with regulators and stakeholders.
The Challenge
The Challenge: Coordinating Cold Chain Crisis Response
Pharmaceutical cold chains are uniquely vulnerable. Products must remain within narrow temperature ranges, every action must comply with GDP and cGMP standards, and delays can put both patients and revenue at risk. Yet most organizations still depend on siloed systems and manual processes. When crises occur, coordination breaks down — responses are delayed, inconsistent, and difficult to document for regulators.
Key Challenge Areas
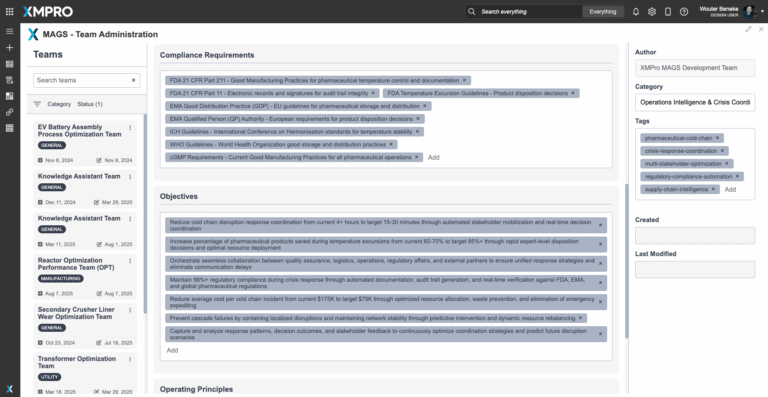
- Temperature Excursions: Even brief deviations can compromise entire batches. With annual global losses exceeding $35B, current monitoring systems often detect issues but cannot coordinate rapid resolution.
- Fragmented Coordination: Manufacturers, logistics providers, distributors, and healthcare organizations rely on calls and emails. Response times stretch beyond hours instead of the 15–30 minutes needed to prevent product loss.
- Inconsistent Product Disposition: Acceptance or rejection decisions depend on which human expert is available, leading to both unnecessary rejections and unsafe acceptances.
- Regulatory Documentation Gaps: Manual processes during crises make it difficult to maintain GDP/cGMP documentation, increasing audit failures, penalties, and rejected insurance claims.
- Technology and Organizational Silos: IoT sensors, ERP, quality systems, and communication platforms rarely integrate, creating blind spots. Escalation rules and decision authority remain unclear during emergencies.
The Compound Impact
These gaps drive direct financial losses, product recalls, and higher insurance costs. They damage strategic supplier and distributor relationships, slow market access, and erode trust with regulators and patients. Traditional monitoring and reporting tools cannot solve this — what’s needed is governed, intelligent coordination across all cold chain stakeholders to respond consistently, transparently, and within compliance.
XMPro Pharmaceutical Cold Chain Crisis Response MAGS Team
From Reactive Firefighting to Governed, Proactive Response
Pharmaceutical cold chains fail less from missing data than from delayed decisions. Temperature excursions, documentation delays, and logistics disruptions demand time-critical coordination under strict GDP and cGMP standards. Traditional tools optimize tasks in isolation, but cannot balance compliance, safety, and efficiency in real time.
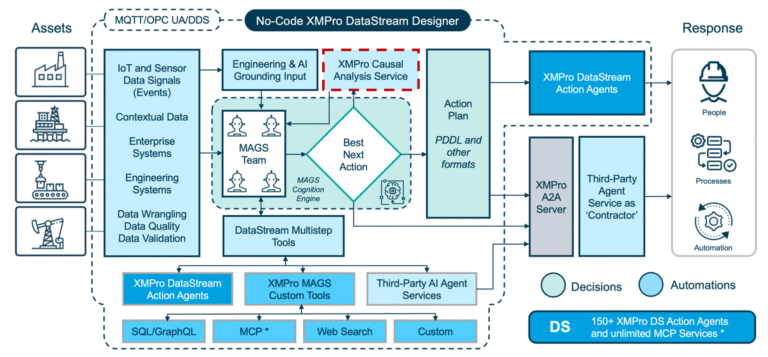
XMPro’s Cold Chain Crisis Response MAGS Team addresses this gap. Each specialized agent — spanning Quality Assurance, Supply Operations, Logistics, Compliance, and Crisis Coordination — operates with bounded autonomy, reasoning through brain-inspired cognitive cycles and collaborating via MAGS consensus protocols. Together, they detect risks, weigh trade-offs, and coordinate corrective action in minutes rather than hours.
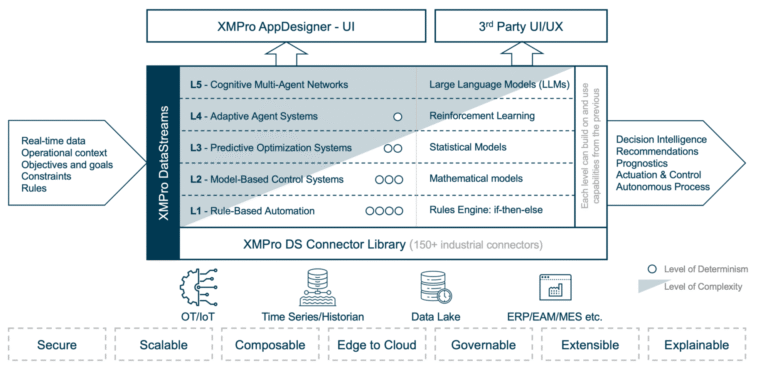
APEX, XMPro’s Agentic Platform Experience, functions as the control room for lifecycle management. It allows organizations to design, deploy, monitor, and improve agents over time, ensuring continued alignment with regulatory policy and business priorities.
The result: a cold chain that coordinates itself — protecting patient safety, minimizing losses, and maintaining compliance with full transparency and trust.
Key Features
Key Features of the Cold Chain Crisis Response MAGS Team
Multi-Agent Collaboration
Five specialized agents — Quality Assurance, Supply Continuity, Logistics Optimization, Regulatory Compliance, and Crisis Detection & Escalation — collaborate through XMPro’s consensus mechanism to detect risks, evaluate trade-offs, and coordinate corrective action in governed, auditable ways.
Real-Time Anomaly Detection
The team continuously ingests IoT sensor data, shipment telemetry, and compliance records, detecting deviations such as temperature excursions, transport delays, or documentation gaps within minutes.
Governed Escalation Paths
Urgent risks are surfaced with urgency weighting and routed through structured escalation protocols. Depending on severity, issues are resolved automatically, raised to human oversight, or communicated to external partners — always within compliance guardrails.
Parametric Configuration
Organizations can configure operating thresholds (e.g., acceptable delay times, excursion tolerances, compliance priorities) so the team aligns with business strategy, patient safety requirements, and regulatory standards.
Digital Twin of Cold Chain Operations
The team maintains a living digital twin that integrates product stability data, lane performance, inventory positions, and incident histories, enabling predictive interventions and scenario testing.
Objective Function Optimization
Team decisions balance competing priorities: patient safety, regulatory compliance, continuity of supply, operational efficiency, and financial risk. No single goal dominates, ensuring holistic crisis response.
Progressive Autonomy
Start with advisory mode (alerts and playbooks), move to supervised execution (automated recommendations requiring approval), and progress to governed autonomy for low-risk responses — always with transparency and audit trails.
MAGS Team Composition
Meet Your Cold Chain Crisis Response Specialists
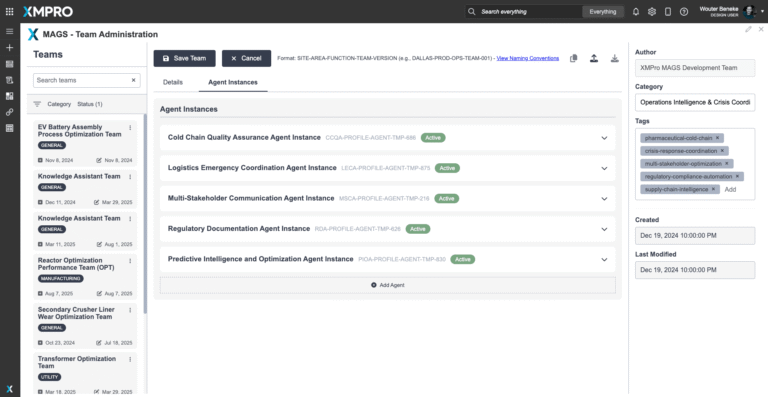
Cold Chain Quality Assurance Agent
(Integrity & Stability Specialist)
Key Expertise: Monitors product stability, temperature excursions, and handling deviations across the chain.
Team Contribution: Ensures patient safety by validating quality standards and flagging risks before they escalate.
Supply Continuity Agent
(End-to-End Coordination Specialist)
Key Expertise: Oversees supplier performance, inventory levels, and lane status
Team Contribution: Maintains continuity of supply by identifying bottlenecks and coordinating replenishment.
Logistics Optimization Agent
(Efficiency & Routing Specialist)
Key Expertise: Optimizes transport routes, cold storage utilization, and distribution flows.
Team Contribution: Minimizes cost and delay while safeguarding product integrity in transit.
Regulatory Compliance Agent
(Policy & Documentation Specialist)
Key Expertise: Tracks GDP/cGMP compliance, manages documentation, and validates regulatory alignment.
Team Contribution: Reduces audit risk and ensures every action is traceable, compliant, and report-ready.
Crisis Detection & Escalation Agent
Crisis Detection & Escalation Specialist
Key Expertise: Detects disruptions, escalates urgent risks, and coordinates rapid cross-agent interventions.
Team Contribution: Surfaces urgent risks, assigns urgency weightings, and ensures they are addressed within the team’s consensus framework
Team Objective Function
Team Objective Function
Collective Success Metrics
Each agent has its own domain-specific objective function, but success is only meaningful when measured at the team level. The Cold Chain Crisis Response MAGS Team integrates these objectives into a unified framework that ensures patient safety, regulatory compliance, and continuity of supply are never compromised by cost or speed pressures.
Individual vs. Team Objectives
- Quality Assurance Agent: Safeguards product integrity through stability and excursion monitoring.
- Supply Continuity Agent: Maintains uninterrupted flow of inventory and supplier reliability.
- Logistics Optimization Agent: Ensures timely, efficient routing while minimizing risk of excursions.
- Regulatory Compliance Agent: Validates that all responses remain GDP/cGMP compliant and audit-ready.
- Crisis Detection & Escalation Agent: Detects anomalies early, prioritizes urgent risks, and drives governed escalation.
Team-Level Metrics
- Product Integrity → % of shipments maintained within validated stability ranges; excursion response time.
- Regulatory Compliance → Audit readiness score, documentation accuracy, % adherence to GDP/cGMP standards.
- Continuity of Supply → On-time delivery %, disruption recovery speed, successful lane handovers.
- Resilience → Crisis resolution time, % of crises mitigated before product loss, escalation accuracy.
- Efficiency → Cost per unit shipped, % reduction in wastage, optimized lane utilization.
Why It Matters
The value of the Crisis Response Team is not just faster incident handling — it’s consistent, explainable, and compliant outcomes. By embedding integrity, compliance, and resilience into the team’s shared objective function, XMPro ensures crises are managed in ways that protect patients, maintain trust with regulators, and preserve business continuity.
Individual Agent Contributions
While collective success is measured at the team level, each agent brings unique expertise that contributes to safe, compliant, and resilient cold chain operations. Together, their complementary roles ensure risks are detected early, responses are coordinated, and outcomes remain explainable and auditable.
Cold Chain Quality Assurance Agent — Protects Product Integrity by continuously validating stability conditions (temperature, humidity, handling). Flags deviations in real time and coordinates corrective action to prevent compromised product reaching patients.
Supply Continuity Agent — Secures Uninterrupted Flow of Medicines by balancing supplier reliability, inventory positioning, and distribution handovers. Identifies bottlenecks and activates contingency supply options to maintain service levels under pressure.
Logistics Optimization Agent — Delivers Efficient & Reliable Distribution by optimizing transport routes, storage allocation, and capacity utilization. Minimizes delays and costs while safeguarding against temperature excursions in transit.
Regulatory Compliance Agent — Ensures Governed Operations by embedding GDP/cGMP standards into every action. Validates that decisions are fully compliant, generates audit-ready documentation, and reduces risk of penalties or rejected claims.
Crisis Detection & Escalation Agent — Strengthens Resilience by detecting anomalies early, applying urgency weighting, and recommending escalation pathways. Helps the team prioritize time-critical risks and mobilize coordinated interventions in minutes to hours.
Compound Benefits
When Quality, Supply, Logistics, Compliance, and Crisis Escalation agents work together, their combined impact creates a cold chain that is safer, faster, and more resilient than any one function operating in isolation.
End-to-End Integrity Assurance
By connecting quality monitoring with logistics optimization and supply continuity, the team safeguards product stability from manufacturer to patient. Excursions are not only detected, but also corrected in time to prevent loss.
Coordinated Compliance
Every intervention is automatically validated against GDP/cGMP requirements. Documentation, audit trails, and escalation records are created in real time, reducing compliance gaps and audit risks.
Resilient Crisis Response
Early anomaly detection combined with urgency weighting ensures crises are escalated and resolved in minutes to hours. The team contains disruption before it cascades, protecting both patients and financial outcomes.
Optimized Efficiency Under Pressure
Route optimization, supply balancing, and crisis protocols converge to reduce waste, lower operational costs, and maintain service continuity — even during severe disruptions.
Shared Learning & Continuous Improvement
Every incident, intervention, and outcome is logged into the team’s memory. Lessons learned from one disruption strengthen the whole cold chain, improving preparedness and decision-making over time.
Team Dynamics Summary
Communication Protocol
Continuous Data Sharing
Agents exchange validated data across the cold chain every few minutes, with immediate broadcasts when exceptions occur. Temperature deviations, sensor alerts, or handling delays are shared instantly with the full team, ensuring no single risk is siloed.
Contextualized Alerts
Rather than raw sensor values, agents provide structured updates in terms of risk impact — for example, “Temperature drift of 2.5°C detected for Shipment X; stability window exceeded.” This ensures communication remains actionable for other agents and human stakeholders.
Priority Escalations
Critical events — such as cold chain breaches, supplier non-compliance, or shipment delays for high-value medicines — trigger team-wide coordination within minutes, enabling proactive intervention before product loss occurs.
Human-in-the-Loop Integration
Agents communicate through the same channels used by operations and compliance teams — Microsoft Teams, Slack, email, and dashboards. All escalations and recommendations include reasoning paths and regulatory context, allowing humans to validate or intervene as needed.
External Interfaces
Where appropriate, agents interact with logistics providers, suppliers, and regulatory partners through governed interfaces (APIs, EDI, structured email). This ensures external stakeholders receive consistent, timely communication aligned with compliance protocols.
Governed Collaboration
All communications are logged, auditable, and governed under XMPro’s architecture, ensuring traceability and policy compliance. This provides confidence that every cold chain decision and alert is explainable and defensible during audits or investigations.
Decision Framework
Domain-Weighted Authority
Each agent has defined authority within its domain — quality, compliance, supply operations, network optimization, or crisis response. When a decision is primarily within one domain (e.g., a stability risk), that agent proposes the action, while other agents provide supporting perspectives.
Collaborative Consensus
For cross-functional trade-offs, such as whether to re-route a shipment after a delay, agents contribute domain-specific reasoning. The MAGS consensus mechanism evaluates service, compliance, cost, and risk impacts, ensuring the action taken optimizes patient safety and regulatory integrity alongside operational efficiency.
Regulatory Alignment
The Regulatory Compliance Agent ensures all decisions remain within GxP, FDA, EMA, and WHO guidelines. This prevents operational expediency from undermining compliance obligations, protecting both patient safety and organizational reputation.
Human-in-the-Loop Oversight
High-impact decisions — such as product release after a temperature excursion, discarding shipments, or invoking crisis protocols — are automatically escalated to human managers. Escalation packages include alternative options, quantified risk assessments, agent reasoning paths, and recommended actions.
Auditability and Traceability
Every decision is logged with contributing agents, reasoning paths, and weighted trade-offs. This ensures complete transparency for regulatory audits and supports continuous improvement of cold chain processes.
Conflict Resolution
Safety-First Resolution Hierarchy
Conflicts are resolved using a governed hierarchy of priorities: Patient Safety → Regulatory Compliance → Service Continuity → Operational Efficiency → Cost. This ensures that no operational or financial objective ever overrides the need to protect patients and maintain compliance with regulatory requirements.
Structured Trade-Off Evaluation
When agents propose conflicting actions — such as rerouting a shipment versus maintaining lowest transport cost — the system evaluates each option against safety, compliance, service, and cost dimensions. Outcomes are scored to highlight the safest and most compliant pathway that also considers service and efficiency.
Collaborative Consensus Mechanism
Rather than voting, agents contribute domain-specific insights: the Quality Agent quantifies product integrity risks, the Regulatory Agent enforces compliance guardrails, the Supply Operations Agent measures fulfillment impact, and the Network Optimization Agent considers routing alternatives. These insights are synthesized through the MAGS consensus algorithm into a unified resolution.
Escalation for Critical Trade-Offs
When a decision involves significant patient impact, compliance uncertainty, or high-value product risk, the system escalates to human oversight. Escalation packages include the following:
- Options evaluated by the agents
- Quantified patient safety and compliance risks
- Service and cost trade-offs
- Agent reasoning paths and recommended resolution
Learning from Resolution
All conflicts and their outcomes are logged. Agents continuously learn from these decisions, improving their ability to manage trade-offs and reducing future conflicts — creating a progressively safer, more reliable cold chain.
Load Balancing
Dynamic Monitoring Intensity
Agents automatically adjust their monitoring cycles and communication frequency based on current conditions. For example, during a heatwave, the Quality Assurance Agent increases temperature monitoring frequency across shipments, while the Network Optimization Agent intensifies route evaluations for at-risk lanes.
Condition-Based Adaptation
When customs delays or regulatory checks occur, the Regulatory Compliance Agent heightens oversight of release requirements, while the Crisis Response Agent elevates coordination activity. The Supply Operations Agent simultaneously reassesses allocation priorities to protect patient-critical deliveries.
Collaborative Attention Allocation
The team redistributes focus dynamically across domains:
• Temperature excursions → Quality Assurance Agent elevates monitoring; Crisis Response Agent prepares mitigation pathways.
• Regulatory hold-ups → Regulatory Compliance Agent increases supervision, supported by Network Optimization for alternative flows.
• Disruption events → Crisis Response Agent raises coordination frequency, while all agents share updates to preserve service continuity.
• Demand spikes → Supply Operations Agent intensifies allocation monitoring, with financial and compliance checks engaged where relevant.
Resilient Workload Distribution
During high-stress periods, processing load is distributed across the team — ensuring no single agent becomes a bottleneck. Each agent focuses on its domain expertise while contributing to coordinated team consensus.
Adaptive Equilibrium
As conditions normalize, agents return to baseline monitoring and communication patterns, maintaining a balanced and efficient cold chain oversight without overloading any single domain.
Escalation Paths
Threshold-Based Escalation
Cold chain operations face zero tolerance for certain risks — particularly temperature excursions, product integrity concerns, and regulatory non-compliance. The team escalates when defined thresholds are breached, such as:
• Patient-critical product at risk of spoilage
• Regulatory non-compliance with release protocols
• Multi-country disruption impacting continuity of supply
• Financial exposure beyond tolerance levels
• Strategic partner or public health commitments at risk
Progressive Autonomy
Organizations can control how escalations are handled based on maturity and confidence in the agent team:
• Advisory Mode → Agents generate alerts and recommended actions, but all external communications and interventions require human approval.
• Sub-Autonomous Mode → Routine responses (e.g., triggering backup monitoring, adjusting carrier allocation) are executed automatically, while high-impact issues are escalated.
• Autonomous Mode → Agents execute governed actions within safe boundaries (e.g., activating an alternate cold storage facility) and escalate only when risks exceed confidence or policy thresholds.
Structured Escalation Packages
When escalation occurs, human managers and regulators receive structured packages that include the following:
• Options evaluated and discarded
• Quantified impact on patients, compliance, and costs
• Reasoning paths and supporting evidence
• Recommended resolution with time-critical context
Integrated Communication
Escalations are delivered through existing enterprise and compliance channels — Microsoft Teams, Slack, email, regulatory reporting systems — and can extend to suppliers, logistics partners, or distributors when their intervention is required.
Learning & Trust Building
Every escalation is logged, reviewed, and used to improve future response. As trust in the team’s performance grows, organizations can safely increase autonomy thresholds while maintaining patient safety, compliance, and transparency.