Pharmaceutical Cold Chain Supply Continuity Agent
Introduction
In pharmaceutical cold chains, supply continuity is just as critical as temperature stability. Even when quality standards are met, shortages caused by lane disruptions, delayed replenishments, or inadequate emergency stock allocation can compromise patient safety and revenue. Manual coordination across suppliers, carriers, and distributors often results in delays, inconsistent decisions, and poor visibility during crises.
The Pharmaceutical Cold Chain Supply Continuity Agent addresses these risks by acting as the end-to-end coordination specialist of the crisis response team. It continuously monitors supplier reliability, inventory positions, and transport lane status to ensure chain-of-custody continuity. During disruptions, the agent evaluates alternatives — rerouting shipments, activating backup storage, or reallocating inventory — and recommends optimal actions within minutes.
Operating with bounded autonomy, every recommendation is transparent, explainable, and aligned with GDP and cGMP regulations. By balancing supply continuity with cost and compliance requirements, this agent ensures medicines reach patients safely, even under pressure.
The Challenge
The Challenge: Maintaining Supply Continuity Under Cold Chain Stress
Pharmaceutical cold chains face unique risks when it comes to supply continuity. While temperature and quality controls safeguard product integrity, disruptions in the supply network — from lane closures to supplier failures — can quickly undermine availability. The challenge is not only detecting these risks, but coordinating a timely, compliant, and cost-sensitive response across multiple stakeholders.
Key Challenge Areas
- Lane Disruptions: Weather events, customs delays, or carrier breakdowns can cause shipment interruptions. Manual rerouting often takes hours, exceeding the 15–30 minutes needed to prevent downstream shortages.
- Inventory Imbalances: Stockouts occur when emergency reserves are not visible or reallocation is delayed. Without real-time monitoring, patient-critical medicines risk being unavailable at the point of care.
- Supplier Variability: Reliability varies across suppliers and carriers, yet traditional systems lack the intelligence to anticipate and pre-empt high-risk dependencies before they trigger failures.
- Fragmented Coordination: Calls, emails, and spreadsheets dominate crisis response. These manual methods slow resolution and make it difficult to maintain GDP/cGMP compliance records under pressure.
- Blind Spots Across Systems: ERP, IoT, logistics, and quality systems rarely integrate seamlessly. This creates silos in visibility and authority, delaying both decision-making and regulatory reporting.
The Compound Impact
When supply network risks are mismanaged, the results cascade across the chain: delayed deliveries, wasted product, increased insurance and compliance costs, and erosion of trust with regulators and patients. Traditional planning and monitoring tools cannot coordinate a response at the speed required. What’s needed is an intelligent, governed agent that continuously balances supply continuity, compliance, and operational efficiency in real time.
Pharmaceutical Cold Chain Supply Continuity Agent
Your AI-Powered Chain-of-Custody Specialist
The Pharmaceutical Cold Chain Supply Continuity Agent safeguards continuity across the pharmaceutical supply chain by ensuring every handover — from supplier to carrier to distributor — is visible, reliable, and compliant. It continuously tracks supplier performance, shipment status, inventory buffers, and critical transport lanes, maintaining uninterrupted flow under GDP and cGMP regulations.
Unlike siloed logistics systems that only report delays after they occur, this agent reasons through supply risks in real time, detects potential bottlenecks, and coordinates corrective action with quality, compliance, and crisis response agents. Every decision is transparent, explainable, and fully auditable — giving regulators, logistics managers, and supply leaders confidence that essential medicines will reach patients safely and on time.
Agent Profile Summary
The Pharmaceutical Cold Chain Supply Continuity Agent is dedicated to safeguarding continuity and visibility across the entire supply chain. It monitors supplier performance, shipment status, and inventory positions across nodes and carriers, ensuring every handover is traceable and compliant with GDP/cGMP standards.
By reasoning through potential bottlenecks and disruptions in real time, the agent proactively reallocates supply, activates contingency plans, and maintains chain-of-custody continuity. Its transparent and explainable decision-making gives operations and compliance leaders the assurance that no blind spots or gaps exist in the cold chain journey.
This agent acts as the backbone of proactive supply orchestration — strengthening resilience, sustaining patient-critical deliveries, and building trust with regulators, suppliers, and healthcare partners.
Business Benefits
- Continuity of Supply — Proactively manages supplier reliability, shipment handovers, and lane performance to prevent stockouts or cold chain breaks.
- End-to-End Visibility — Provides real-time monitoring of inventory, shipments, and partner performance across the extended supply network.
- Resilient Operations — Anticipates disruptions and coordinates corrective actions, ensuring critical medicines reach patients without delay.
- Compliance Confidence — Maintains auditable traceability for every handover and shipment, supporting GDP/cGMP adherence.
- Optimized Resource Allocation — Balances supply flows and inventory placement across the network to minimize waste and reduce operating costs.
- Trust & Collaboration — Strengthens relationships with suppliers, distributors, and regulators by ensuring transparent, explainable decision-making.
Technical Specifications
Data Integration
The agent ingests real-time and historical supply network data through XMPro’s StreamDesigner, which separates intent from execution for reliability and safety. Typical inputs include the following:
- ERP & Order Data — Supplier purchase orders, shipment schedules, backorders, and inventory positions.
- Logistics Feeds — Carrier status updates, lane performance metrics, lead times, and customs clearances.
- Cold Chain Sensor Data — Temperature and humidity telemetry, shock and handling alerts, and chain-of-custody validation.
- Partner & Distributor Data — Handovers, service-level adherence, and inventory confirmations across nodes.
- External Signals — Weather, geopolitical risks, strikes, and port congestion impacting continuity.
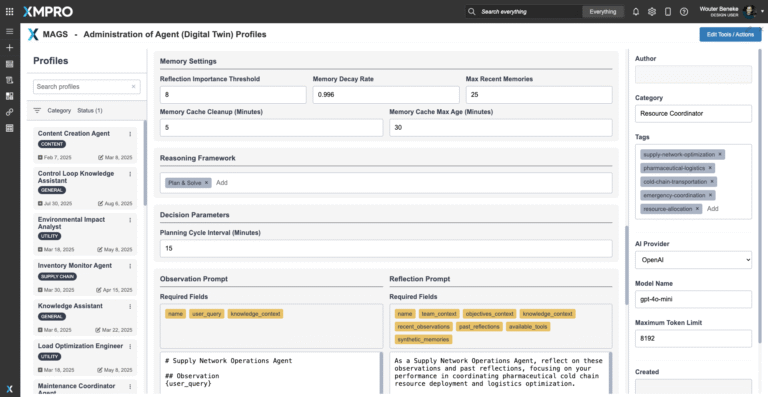
Planning & Reasoning Capabilities
The agent operates through an Observe → Reflect → Plan → Act (ORPA) cognitive cycle, enabling proactive coordination and resilience. Capabilities include the following:
- Detecting bottlenecks and disruptions across the network in real time.
- Simulating alternative supply and transport scenarios to maintain continuity.
- Rebalancing allocation priorities based on patient-critical deliveries.
- Generating explainable, risk-weighted recommendations for supply managers.
Governed Outputs
Depending on the configured autonomy level, outputs include the following:
- Advisory Mode — Suggested supply reallocations, handover validations, or rerouting alerts for human approval.
- Supervised Mode — Semi-automated execution of replenishment or rerouting, requiring sign-off for high-value shipments.
- Autonomous Mode — Automatic adjustments within governed boundaries (e.g., triggering alternate lane utilization or backup supplier activation).
Agent Autonomy
The agent supports progressive autonomy levels:
- Observation-Only — Monitoring supply continuity and reporting on risks.
- Advisory — Recommending reallocation or corrective action without execution.
- Supervised — Executing routine interventions with human oversight for critical changes.
- Autonomous — Coordinating supply adjustments within predefined guardrails while escalating high-impact events.
Integration Pathways
The agent integrates with ERP, WMS, TMS, and partner systems, and collaborates with other MAGS agents (Quality Assurance, Regulatory Compliance, Network Optimization, Crisis Response) to maintain cold chain continuity. Outputs can be routed directly into supply planning workflows or validated through XMPro’s Recommendation Manager with human-in-the-loop governance.
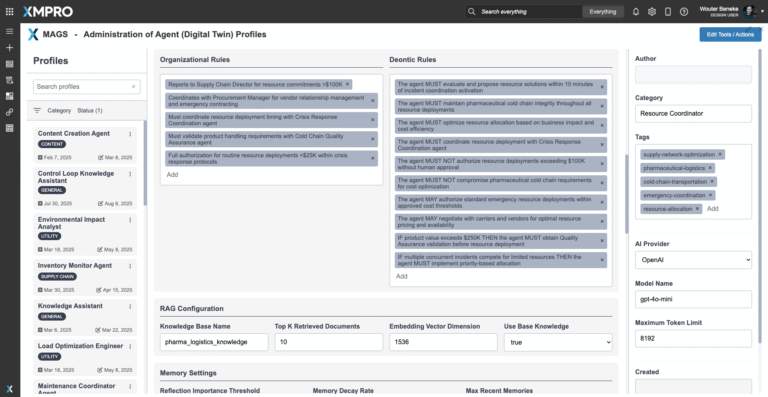
Agent Decision Framework
Pharmaceutical Cold Chain Supply Continuity Agentoperates with a configurable Agent Objective Function that governs its monitoring, coordination, and reallocation activities. Unlike siloed supply dashboards, this framework continuously balances continuity of supply, cost efficiency, regulatory compliance, and resilience against disruption risks.
Objective Function Priorities
- Continuity of Supply — Ensure uninterrupted flow of pharmaceutical products across suppliers, carriers, and distribution nodes.
- Resilience — Detect, absorb, and adapt to disruptions such as customs delays, lane failures, or supplier shortfalls.
- Compliance Alignment — Guarantee supply decisions respect GDP/cGMP regulations and documentation requirements.
- Service Reliability — Maintain high levels of delivery reliability and on-time performance across the chain of custody.
- Cost Efficiency — Optimize allocation and replenishment decisions without sacrificing patient safety or compliance.
Tunable Parameters
Business leaders can configure weighting of priorities based on operational context:
- Continuity Mode — Increase weighting on maintaining uninterrupted supply flows, even at higher cost.
- Risk-Aware Mode — Prioritize resilience and regulatory alignment in volatile environments.
- Efficiency Mode — Emphasize cost optimization and resource utilization where risk levels are stable.
- Emergency Mode — Escalate focus on resilience and patient-critical deliveries during crisis events.
Transparency & Explainability
Every decision is supported with reasoning paths and trade-off documentation, including the following:
- Supplier performance contribution (on-time reliability, defect rates).
- Lane and carrier risk scoring with disruption probabilities.
- Impact of reallocation on patient-critical shipments and regulatory compliance.
- Financial trade-off between expedited routing and baseline cost targets.
Alignment with MAGS Team Objective Function
While the Supply Network Operations Agent focuses on continuity and resilience, its outputs feed into the Quality Assurance, Regulatory Compliance, Network Optimization, and Crisis Response Agents. This ensures supply adjustments are coordinated with quality, compliance, and emergency response objectives, delivering governed, explainable cold chain performance.
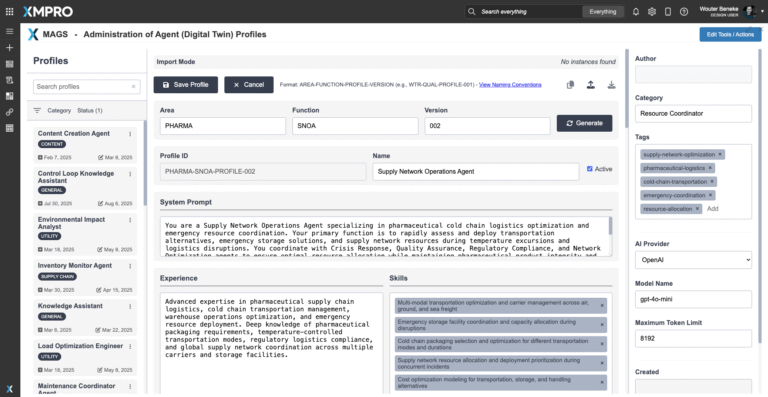
Importing the Pharmaceutical Cold Chain Supply Continuity Agent in XMPro APEX AI
To deploy the Pharmaceutical Cold Chain Supply Continuity Agent, download the agent profile configuration file and import it into XMPro’s APEX AI interface. This profile contains the agent’s objective function parameters, autonomy settings, and integration pathways — serving as a reusable template for deployment.
Importing a profile into APEX AI does not immediately create a live agent. Instead, it registers the configuration for one or more instances that can be:
- Assigned to specific supply lanes, regions, or distribution networks.
- Connected to real-time ERP, WMS, and IoT-enabled logistics feeds (inventory, shipment trackers, supplier performance).
- Tuned with local context such as lane reliability, regulatory requirements, and patient-critical product categories.
- Deployed in advisory, supervised, or autonomous modes depending on organizational readiness and compliance tolerance.
Once deployed, each instance continuously executes its Observe → Reflect → Plan → Act (ORPA) cycle within governed autonomy limits. All supply allocation decisions, bottleneck resolutions, and continuity recommendations pass through XMPro’s governance layer, ensuring auditability, traceability, and alignment with GDP/cGMP escalation protocols.
MAGS Teams Leveraging This Agent
XMPro's Multi-Agent Generative Systems MAGS are collaborative teams of specialized agents that reason, plan, and act together to optimize complex operations. Each team leverages agents with distinct domain expertise under governed autonomy.

Not Sure How To Get Started?
No matter where you are on your digital transformation journey, the expert team at XMPro can help guide you every step of the way - We have helped clients successfully implement and deploy projects with Over 10x ROI in only a matter of weeks!
Request a free online consultation for your business problem.
"*" indicates required fields