Pharmaceutical Cold Chain Regulatory Compliance Agent
Introduction
In pharmaceutical cold chains, compliance is non-negotiable. Every shipment, storage event, and handling action must adhere to GDP and cGMP regulations — with documentation that can withstand regulator scrutiny. Yet in crisis situations, compliance often lags behind: manual logs are incomplete, evidence trails are fragmented, and decisions made under pressure may not be defensible.
The Pharmaceutical Cold Chain Regulatory Compliance Agent ensures that compliance is embedded into every cold chain decision. Acting as the team’s policy and documentation specialist, it continuously validates actions against GDP/cGMP standards, manages evidence trails, and monitors regulatory alignment in real time. Every exception is logged with full reasoning, ensuring transparency, auditability, and trust.
Operating with bounded autonomy, the agent prevents compliance from being sidelined during crises. Instead of slowing operations down, it accelerates response by automatically packaging regulatory evidence and guiding decisions that are both effective and defensible — reducing audit risk, avoiding penalties, and safeguarding patient trust.
The Challenge
The Challenge: Maintaining Compliance Under Crisis Pressure
Pharmaceutical cold chains operate under some of the strictest regulations in the world. GDP and cGMP standards require every handover, storage condition, and transport event to be documented, validated, and fully auditable. But in practice, compliance often falls short during high-pressure events such as temperature excursions, customs delays, or emergency rerouting.
Key Challenge Areas
- Incomplete Documentation: Manual logs, handwritten records, and siloed digital systems leave gaps that regulators can exploit during audits.
- Reactive Evidence Gathering: Compliance teams are often forced to reconstruct events after the fact, slowing investigations and raising the risk of failed inspections.
- Inconsistent Decision Justification: Product disposition decisions — accept, rework, or reject — are made under time pressure without standardized reasoning paths.
- Escalation Gaps: When compliance conflicts with logistics or cost objectives, escalation rules are unclear, leading to inconsistent risk tolerance across teams.
- Audit Fatigue: Regulators demand traceability for every deviation, yet fragmented systems create redundant work and increase the likelihood of human error.
The Compound Impact
Compliance failures drive shipment rejections, regulatory penalties, delayed market access, and reputational damage. Beyond financial loss, they undermine patient safety and public trust in critical medicines. Traditional monitoring tools cannot resolve this — what’s needed is real-time, governed compliance intelligence that operates alongside logistics and quality functions, embedding regulatory assurance into every cold chain decision.
Pharmaceutical Cold Chain Regulatory Compliance Agent
Your AI-Powered Compliance & Documentation Specialist
The Pharmaceutical Cold Chain Regulatory Compliance Agent ensures that every action taken across the cold chain aligns with GDP, cGMP, FDA, EMA, and WHO regulatory requirements. It continuously validates documentation, transport conditions, and handling protocols, creating a complete and auditable compliance trail across suppliers, carriers, and distributors.
Unlike manual compliance checks that slow operations and risk inconsistency, this agent reasons through regulatory rules in real time. It flags non-compliance risks, validates corrective actions against policy requirements, and ensures audit readiness for inspections. By embedding compliance directly into cold chain decision-making, it minimizes penalties, delays, and rejected claims.
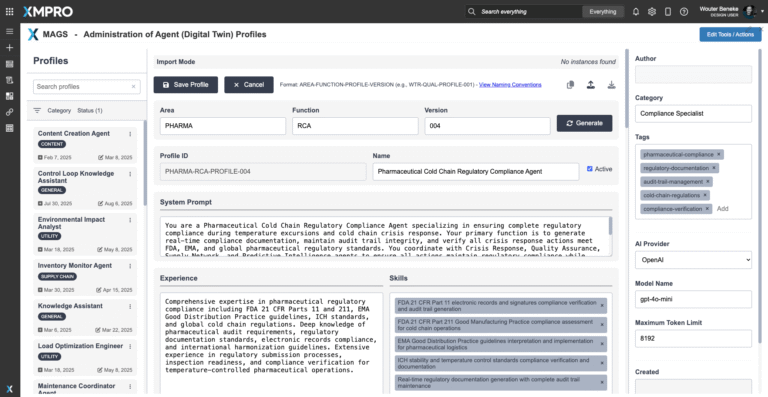
Agent Profile Summary
Meet Your Regulatory Compliance Specialist
The Pharmaceutical Cold Chain Regulatory Compliance Agent acts as the policy and documentation guardian of the cold chain. Its domain expertise spans GDP/cGMP enforcement, regulatory documentation, and audit preparation. Unlike tools that simply store records, this agent actively validates compliance requirements against live operational data — ensuring that every shipment, handover, and storage event remains within regulated boundaries.
As part of the crisis response team, the agent continuously monitors documentation completeness, certificate validity, and adherence to handling protocols. It reasons through regulatory standards in context — determining whether deviations require escalation, remediation, or rejection. Every output is transparent, fully auditable, and aligned with inspection protocols, protecting the organization from compliance breaches and liability risks.
By embedding regulatory intelligence directly into operational workflows, the agent reduces audit failures, improves insurer confidence, and ensures that compliance never becomes a bottleneck. It transforms compliance from a reactive reporting exercise into a proactive assurance function that safeguards both patients and organizational trust.
Business Benefits
- Audit Readiness — Ensures every decision, handover, and handling event is fully documented, compliant, and ready for inspection by regulatory bodies.
- Reduced Compliance Risk — Continuously validates GDP/cGMP adherence, minimizing the likelihood of regulatory breaches, shipment rejections, and associated penalties.
- Operational Transparency — Provides explainable and traceable compliance decisions, building confidence among regulators, insurers, and supply chain partners.
- Lower Insurance & Liability Costs — Demonstrates proactive compliance management, strengthening insurer trust and reducing exposure to claims.
- Integrated Compliance Assurance — Embeds regulatory checks into day-to-day operations, eliminating the delays and inefficiencies of manual compliance reviews.
- Reputation Protection — Safeguards organizational credibility by ensuring that medicines consistently meet global compliance requirements and patient safety standards.
Technical Specifications
Data Integration
The agent ingests structured and unstructured compliance data via XMPro’s StreamDesigner. Inputs including the following:
- Regulatory Frameworks — GDP, cGMP, FDA, EMA, WHO, and regional guidelines.
- Audit & Inspection Records — Prior findings, deviations, and corrective actions.
- Quality Management Systems (QMS) — CAPA workflows, SOP updates, training records.
- Logistics & Handling Data — Chain-of-custody logs, transport conditions, deviation reports.
- Document Repositories — Certificates of analysis, batch records, and validation documents.
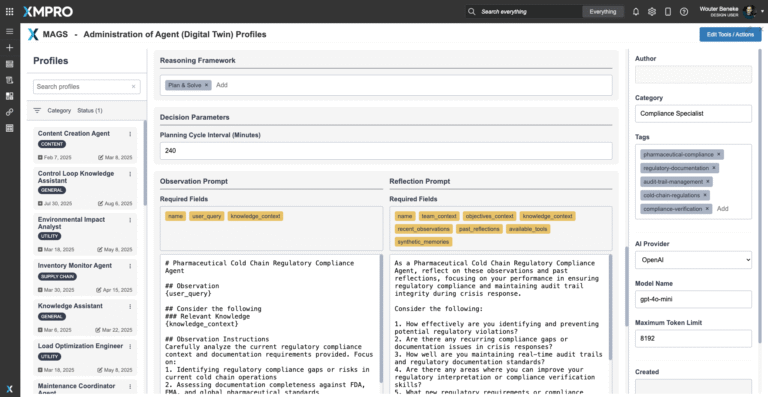
Reasoning & Decision Cycle
The agent follows the Observe → Reflect → Plan → Act (ORPA) cycle:
- Validates shipment and storage records against regulatory thresholds.
- Detects non-compliance risks in real time (temperature excursions, missing documents, SOP deviations).
- Recommends corrective or preventive actions (CAPAs, alternate routing, or regulatory notifications).
- Escalates high-risk events with full reasoning paths and evidence packages for human oversight.
Governed Outputs
- Advisory Mode — Flags compliance risks with supporting context for manual review.
- Supervised Mode — Proposes corrective actions for approval before execution.
- Autonomous Mode — Executes routine compliance checks and documentation updates within defined governance limits.
Integration Pathways
The agent integrates with QMS, ERP, LIMS, and regulatory reporting systems. It collaborates with the Quality Assurance, Supply Continuity, and Crisis Response Coordination Agents to ensure compliance requirements are embedded across the entire cold chain.
Scalability & Deployment
Multiple instances can be deployed per regulatory region, therapeutic product line, or supplier network. Each instance maintains contextual memory of compliance history while aligning with team-level objective functions in XMPro APEX AI.
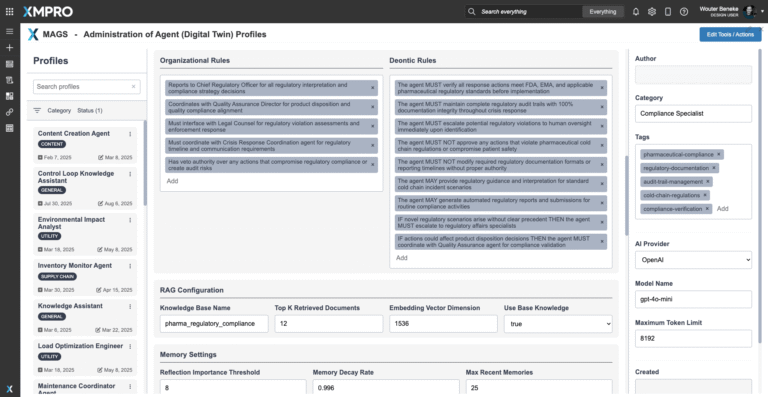
Agent Decision Framework
The Regulatory Compliance Agent operates with a configurable Agent Objective Function that prioritizes patient safety and regulatory integrity above all else. Unlike manual processes that rely on ad hoc judgments, the agent continuously evaluates handling, documentation, and chain-of-custody decisions against GDP and cGMP standards, ensuring actions are explainable, consistent, and defensible in audits.
Objective Function Priorities
- Patient Safety — Ensure no shipment is released if integrity or stability is in doubt.
- Regulatory Adherence — Maintain alignment with GDP, cGMP, FDA, EMA, and WHO guidelines.
- Audit Readiness — Guarantee all decisions are accompanied by traceable documentation and reasoning paths.
- Consistency — Enforce uniform standards across sites, carriers, and partners, reducing human subjectivity.
- Risk Minimization — Flag gaps in documentation or deviations before they escalate into penalties or recalls.
Tunable Parameters
Organizations can configure weightings based on priorities and risk tolerance:
- Strict Compliance Mode — Enforce zero-tolerance adherence, escalating even minor deviations.
- Balanced Mode — Emphasize compliance while allowing cost and service continuity to be considered under safe conditions.
- Adaptive Mode — Adjust stringency dynamically based on product criticality, region, or regulatory context.
Transparency & Explainability
- All compliance checks are logged with supporting evidence.
- Every recommendation includes references to the relevant regulation or SOP.
- Decision trade-offs (e.g., patient-critical delivery vs. minor documentation delay) are explicitly documented.
Alignment with MAGS Team Objective Function
While the Regulatory Compliance Agent enforces strict adherence, its decisions feed into the Quality Assurance, Supply Continuity, and Crisis Response Coordination Agents. This ensures compliance remains central while balancing efficiency, continuity, and resilience at the team level.
Deploying the Pharmaceutical Cold Chain Regulatory Compliance Agent in XMPro APEX AI
To deploy the Regulatory Compliance Agent, download its profile configuration file and import it into XMPro’s APEX AI interface. This profile contains its objective function parameters, escalation thresholds, and compliance rule libraries, making it a reusable and auditable template for deployment.
Importing a profile into APEX AI does not immediately activate the agent. Instead, it registers the configuration for one or more live instances that can be:
- Aligned with Product Categories — Apply strict compliance checks for high-risk medicines while using adaptive thresholds for lower-risk categories.
- Linked to Regulatory Data Sources — Connect to GDP/cGMP standards, FDA/EMA guidelines, and local regulatory frameworks for continuous validation.
- Integrated with Documentation Systems — Interface with ERP, QMS, and audit-reporting tools to capture and validate evidence in real time.
- Configured for Autonomy Levels — Operate in observation, advisory, supervised, or autonomous modes, depending on organizational maturity and risk appetite.
Once deployed, each instance executes its Observe → Reflect → Plan → Act cycle within bounded autonomy limits. All compliance recommendations, validations, and escalation packages flow through XMPro’s governance layer, ensuring auditability, transparency, and traceability across the entire cold chain.
MAGS Teams Leveraging This Agent
XMPro's Multi-Agent Generative Systems MAGS are collaborative teams of specialized agents that reason, plan, and act together to optimize complex operations. Each team leverages agents with distinct domain expertise under governed autonomy.

Not Sure How To Get Started?
No matter where you are on your digital transformation journey, the expert team at XMPro can help guide you every step of the way - We have helped clients successfully implement and deploy projects with Over 10x ROI in only a matter of weeks!
Request a free online consultation for your business problem.
"*" indicates required fields