Pharmaceutical Cold Chain Quality Assurance Agent
Introduction
Pharmaceutical products are highly sensitive to temperature, humidity, and handling conditions. Even short excursions can compromise entire batches, risking patient safety, financial loss, and regulatory penalties. Traditional monitoring systems detect anomalies but leave response fragmented, slow, and difficult to audit under GDP and cGMP requirements.
The Pharmaceutical Cold Chain Quality Assurance Agent addresses this challenge by acting as the integrity guardian of the cold chain. It continuously validates product stability against defined quality thresholds, flags deviations in real time, and ensures corrective actions are traceable and compliant. Operating with governed autonomy, every recommendation is transparent, explainable, and aligned with regulatory standards.
The result: a proactive safeguard that protects product integrity, reduces unnecessary waste, and builds trust with regulators and patients alike.
The Challenge
The Challenge: Protecting Product Integrity in the Cold Chain
Pharmaceutical cold chains operate under strict GDP and cGMP requirements, where even minor deviations can have severe consequences. The integrity of vaccines, biologics, and temperature-sensitive drugs depends on maintaining validated ranges throughout manufacturing, storage, and transport. Yet ensuring this integrity is far more complex than simply recording temperature.
Key Challenge Areas
- Temperature Excursions: Products can lose stability within minutes of exposure. Monitoring systems often raise alerts but cannot ensure timely, coordinated resolution.
- Humidity & Handling Risks: Packaging damage, condensation, or improper handling at transfer points frequently go undetected until quality reviews — often too late to save product.
- Fragmented Oversight: Quality data is siloed across IoT sensors, warehouses, logistics providers, and ERP systems, preventing holistic visibility of product status.
- Inconsistent Product Disposition: Acceptance or rejection decisions depend on which human expert is available, leading to both unnecessary discards and unsafe approvals.
- Regulatory Pressure: Documentation gaps and delayed responses increase audit risk, expose organizations to penalties, and erode trust with regulators and patients.
The Compound Impact
Without a proactive, governed approach, pharmaceutical companies face mounting losses from wasted product, higher insurance costs, and reputational damage. Patient safety can be compromised, and organizations struggle to prove compliance under regulatory scrutiny. What’s needed is a dedicated assurance capability that continuously validates quality in real time, coordinates timely interventions, and makes every action transparent and auditable.
Pharmaceutical Cold Chain Quality Assurance Agent
Your AI-Powered Integrity & Stability Specialist
The Pharmaceutical Cold Chain Quality Assurance Agent safeguards product integrity across the most critical point of the pharmaceutical supply chain: temperature-sensitive transport and storage. It continuously monitors temperature, humidity, and handling conditions, validating every excursion against stability data and regulatory standards such as GDP and cGMP.
Unlike traditional monitoring systems that only raise alerts, this agent reasons through anomalies, determines whether stability windows have truly been breached, and coordinates corrective actions with logistics, compliance, and operations teams. Every decision is transparent, explainable, and fully auditable — giving regulators and quality managers confidence in the integrity of every shipment.
This agent transforms quality assurance from a reactive compliance checkpoint into a proactive intelligence function. By reducing temperature excursion losses, preventing unnecessary product rejections, and ensuring validated compliance, it provides pharmaceutical companies with a reliable foundation for decision-making — protecting patient safety, minimizing financial loss, and strengthening regulatory trust.
Agent Profile Summary
Meet Your Specialist
The Pharmaceutical Cold Chain Quality Assurance Agent is your dedicated safeguard for ensuring that every shipment remains within validated stability ranges. It acts as a digital quality specialist embedded throughout your cold chain operations, combining continuous sensor validation, anomaly detection, and corrective guidance into a single, explainable system.
- Key Expertise: Continuous monitoring of temperature, humidity, and handling conditions across shipments, facilities, and distribution nodes.
- Domain Focus: Ensures every product meets GDP and cGMP quality standards, validating data integrity and stability compliance in real time.
- Unique Strength: Goes beyond raising alarms by reasoning through anomalies, differentiating between transient fluctuations and critical breaches, and recommending evidence-backed actions.
- Core Value: Protects patient safety and company reputation by reducing false rejections, preventing avoidable product loss, and ensuring every decision stands up to regulatory scrutiny.
This agent is the foundation of cold chain trust — proactively validating integrity at every step so that pharmaceutical companies can move from firefighting quality crises to orchestrating safe, compliant, and reliable operations.
Business Benefits
The Pharmaceutical Cold Chain Quality Assurance Agent delivers measurable value by transforming how organizations safeguard product integrity in regulated environments. Its continuous monitoring and explainable decision-making framework not only protect patients but also strengthen financial and operational resilience.
- Reduced Product Loss: Minimizes spoilage and unnecessary batch rejections by detecting and resolving excursions in real time.
- Regulatory Confidence: Ensures every decision is traceable, auditable, and compliant with GDP and cGMP, reducing audit failures and penalties.
- Improved Patient Safety: Guarantees validated product quality across the supply chain, lowering the risk of compromised medicines reaching patients.
- Operational Efficiency: Automates anomaly validation and corrective guidance, reducing firefighting and freeing experts for higher-value work.
- Financial Protection: Cuts insurance claims, recall costs, and write-offs by maintaining stability across shipments and facilities.
- Stronger Partner Trust: Builds confidence with regulators, distributors, and healthcare providers through transparent, explainable quality assurance.
What You Need to Know
Data Integration
The agent ingests real-time and historical cold chain data through XMPro’s StreamDesigner, which separates intent from action for safety and reliability. It handles acquisition, validation, and contextualization of multiple data types, including:
-
IoT Sensor Data temperature, humidity, vibration, light exposure, and shock monitoring across shipments and storage nodes.
-
Calibration Records validation against sensor baselines to eliminate false positives and ensure reliable anomaly detection.
-
Quality & Stability Data batch release specs, product stability profiles, and excursion tolerance thresholds.
-
Logistics & Handling Events carrier scans, lane transitions, dwell times, and handling deviations.
-
Regulatory Standards GDP and cGMP rulesets defining acceptable handling, reporting, and documentation practices.
-
External Conditions weather alerts, regional disruption data, and customs risk indicators that may affect cold chain stability.
All data is synchronized and contextualized before entering the reasoning cycle, ensuring every recommendation is both compliant and explainable.
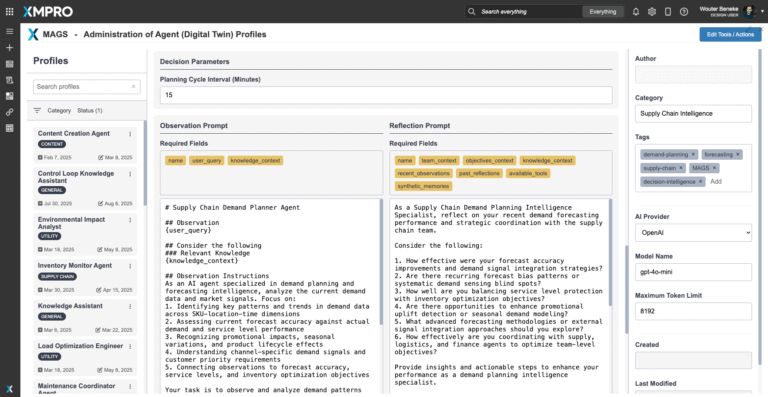
Planning & Reasoning Capabilities
The agent follows a structured Observe → Reflect → Plan → Act (ORPA) cycle, combining anomaly detection, predictive modeling, and compliance rulesets. Its reasoning includes:
-
Detecting and validating temperature excursions in real time.
-
Correlating excursions with handling or logistics events to identify root causes.
-
Recommending corrective actions (reroute, quarantine, activate backup storage) with quantified risk scores.
-
Embedding GDP/cGMP compliance rules into every decision, ensuring audit-readiness.
-
Producing explainable outputs with traceable reasoning paths for regulators and quality managers.
Governed Outputs
Depending on configured autonomy level, outputs include the following:
-
Advisory Mode excursion alerts with recommended actions sent to QA teams for validation.
-
Supervised Mode automatic quarantine or rerouting recommendations executed with human sign-off.
-
Autonomous Mode direct interventions within predefined guardrails (e.g., switching to backup cooling units or cold storage facilities).
All outputs pass through XMPro’s governance layer (APEX AI + StreamDesigner), enforcing bounded autonomy, escalation rules, and audit compliance.
Agent Autonomy
The agent supports progressive autonomy:
-
Observation-Only monitoring and reporting excursions without intervention.
-
Advisory generating corrective action recommendations without execution.
-
Supervised executing low-risk interventions with QA approval on high-impact actions.
-
Autonomous continuous integrity protection actions within regulated boundaries.
Integration Pathways
The agent integrates with IoT platforms, ERP, QMS, LIMS, WMS, and logistics systems, and collaborates with other MAGS agents (Supply, Regulatory, Network Optimization, Crisis Coordination) to ensure product integrity aligns with broader cold chain objectives. Outputs can flow directly into quality workflows or be routed through XMPro’s Recommendation Manager for human-in-the-loop oversight.
Scalability & Deployment
The agent can be deployed across multiple product categories, shipping lanes, and geographies. Each instance maintains its own contextual memory (stability profiles, excursion history, regulatory requirements) while participating in team-level coordination under MAGS. This ensures explainable, safe, and compliant quality assurance at both shipment and enterprise levels.
Agent Decision Framework
The Cold Chain Quality Assurance Agent operates with a configurable Agent Objective Function that governs its monitoring, assessment, and disposition decisions. Unlike static SOP checklists, this framework continuously balances patient safety, regulatory compliance, service continuity, and cost efficiency under dynamic cold chain conditions.
Objective Function Priorities
- Product Integrity — Ensure temperature, humidity, and handling remain within validated ranges; minimize risk of spoilage or potency loss.
- Regulatory Adherence — Guarantee all actions comply with GDP/cGMP requirements and documentation standards.
- Patient Safety — Escalate immediately when risks to end-user safety are detected, regardless of financial trade-offs.
- Disposition Accuracy — Reduce both false rejections and unsafe acceptances by applying evidence-based decision criteria.
- Operational Continuity — Support timely product release and minimize unnecessary disruption while maintaining compliance guardrails.
Deploying the Supply Chain Demand Planner Agent in XMPro APEX AI
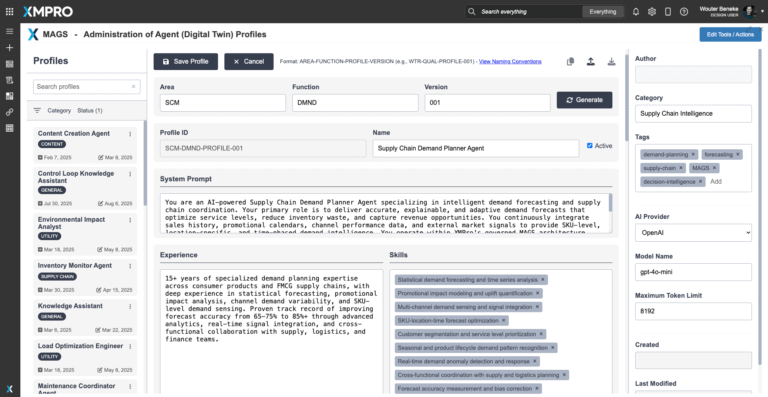
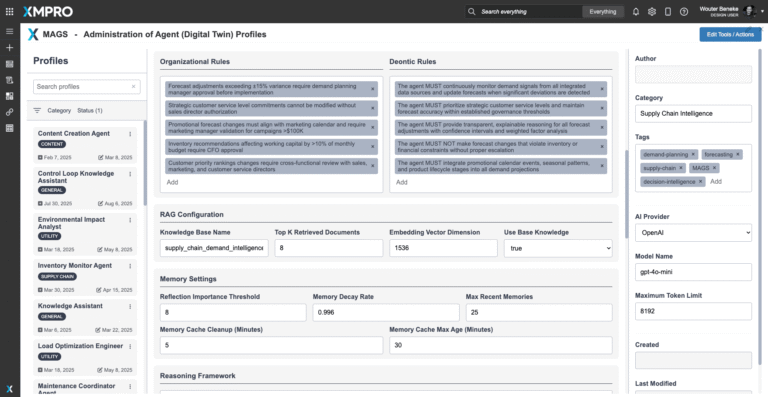
To deploy the Supply Chain Demand Planner Agent, download the agent profile configuration file and import it into XMPro’s APEX AI interface. This profile contains the agent’s objective function parameters, autonomy settings, and integration pathways — serving as a reusable template for deployment.
Importing a profile into APEX AI does not immediately create a live agent. Instead, it registers the configuration for one or more instances that can be
-
Assigned to specific product categories, regions, or channels.
-
Connected to real-time ERP, CRM, and POS data sources.
-
Tuned with local context (e.g., SKU hierarchies, shelf-life constraints, customer segmentation).
-
Deployed as advisory, supervised, or autonomous instances depending on organizational readiness.
Once deployed, each instance continuously executes its Observe → Reflect → Plan → Act cycle within governed autonomy limits. All forecast updates, adjustments, and recommendations pass through XMPro’s governance layer, ensuring auditability, traceability, and compliance with escalation protocols.
MAGS Teams Leveraging This Agent
XMPro's Multi-Agent Generative Systems MAGS are collaborative teams of specialized agents that reason, plan, and act together to optimize complex operations. Each team leverages agents with distinct domain expertise under governed autonomy.

Not Sure How To Get Started?
No matter where you are on your digital transformation journey, the expert team at XMPro can help guide you every step of the way - We have helped clients successfully implement and deploy projects with Over 10x ROI in only a matter of weeks!
Request a free online consultation for your business problem.
"*" indicates required fields