CSTR Regulatory Compliance Agent
Introduction
In pharmaceutical manufacturing, regulatory compliance is not optional—it's the foundation that ensures patient safety, product quality, and business continuity. Yet most CSTR operations struggle with data integrity monitoring, real-time compliance validation, and maintaining the comprehensive documentation required for FDA inspections while managing complex continuous manufacturing processes.
The CSTR Regulatory Compliance Agent represents a breakthrough in pharmaceutical compliance intelligence—an AI-powered specialist that continuously monitors data integrity, validates parameter compliance, maintains comprehensive audit trails, and ensures regulatory readiness 24/7. Unlike traditional compliance systems that operate reactively, this agent proactively identifies compliance risks and ensures every process decision supports cGMP requirements.
Designed specifically for pharmaceutical CSTR operations, this agent serves as your dedicated regulatory guardian, ensuring that process optimization and operational efficiency never compromise compliance standards or patient safety requirements.
The Pharmaceutical Regulatory Compliance Challenge
Pharmaceutical CSTR operations face an unprecedented regulatory complexity that traditional compliance approaches cannot adequately address. Maintaining continuous cGMP compliance while operating sophisticated chemical processes requires simultaneous monitoring of data integrity, parameter compliance, documentation completeness, and audit trail maintenance—all while supporting innovative continuous manufacturing initiatives and process analytical technology implementations.
Where Traditional Compliance Systems Fall Short
- Reactive compliance monitoring: Issues discovered during audits rather than prevented through continuous monitoring, leading to costly investigations and potential regulatory actions.
- Manual documentation gaps: Human operators struggle to maintain complete batch records while managing complex CSTR operations, resulting in data integrity issues and audit findings.
- Fragmented data systems: Compliance information scattered across DCS, MES, LIMS, and paper records, making comprehensive audit trail maintenance nearly impossible.
- Parameter compliance complexity: Hundreds of critical process parameters require continuous monitoring against specification limits, with manual tracking prone to oversight and errors.
- Change control coordination: Process improvements and optimization require extensive regulatory documentation, often delaying implementation of beneficial changes.
- Audit readiness pressure: Regulatory inspections require immediate access to complete documentation, putting enormous pressure on quality and operations teams.
FDA Continuous Manufacturing Compliance Complexity
- Real-time release testing: Continuous operations require real-time quality decisions supported by comprehensive data integrity and process understanding.
- Process analytical technology validation: PAT systems require extensive validation and continuous compliance monitoring to maintain regulatory approval.
- Electronic batch record integrity: Continuous processes generate massive data volumes requiring real-time validation and electronic signature management.
- 21 CFR Part 11 compliance: Electronic records and signatures require continuous monitoring for data integrity, audit trails, and access controls.
- Process validation maintenance: Continuous operations require ongoing process validation and performance qualification documentation.
Data Integrity and Documentation Challenges
- Data completeness verification: Ensuring all required process parameters are recorded, validated, and available for regulatory review requires continuous monitoring across multiple systems.
- Audit trail maintenance: Every process change, parameter adjustment, and operator action must be documented with complete traceability for regulatory inspection.
- Electronic signature integrity: User authentication, authorization levels, and signature validity require continuous monitoring and validation.
- Calibration and validation tracking: Instrument calibration status, validation protocols, and equipment qualification must be continuously monitored and documented.
- Deviation management: Process deviations require immediate identification, investigation, and documentation to maintain cGMP compliance.
- Document version control: Standard operating procedures, batch records, and validation protocols require version control and change tracking.
The Strategic Impact
These compliance challenges create a reactive cycle where regulatory issues are discovered too late for effective prevention, leading to costly investigations, production delays, and potential regulatory actions. Traditional compliance approaches may fail during FDA inspections, result in warning letters or consent decrees, and ultimately compromise patient safety and business continuity. The complexity of continuous manufacturing operations amplifies these risks significantly.
Breaking the Reactive Compliance Cycle
Solving this challenge requires more than better documentation systems or additional quality personnel—it demands an intelligent, proactive, and continuously learning compliance system that combines regulatory expertise with real-time process monitoring, automated documentation, and predictive compliance analysis for comprehensive regulatory intelligence.
XMPro CSTR Regulatory Compliance Agent
Your AI-Powered Data Integrity & Regulatory Documentation Specialist
The CSTR Regulatory Compliance Agent is an autonomous Decision Agent purpose-built for proactive pharmaceutical regulatory compliance and data integrity monitoring. It continuously validates data completeness, monitors parameter compliance, maintains comprehensive audit trails, and ensures regulatory readiness—transforming reactive compliance management into proactive regulatory intelligence that supports both operational excellence and patient safety.
Operating within XMPro's governed Multi-Agent Generative Systems (MAGS) architecture, this agent uses Composite AI to combine regulatory expertise, data validation algorithms, compliance monitoring, and predictive analysis. Unlike reactive compliance systems, it provides real-time regulatory intelligence that prevents compliance issues before they occur while supporting innovative continuous manufacturing and process analytical technology implementations.
Agent Profile Summary
Meet Your New Regulatory Intelligence & Compliance Specialist
The CSTR Regulatory Compliance Agent is a governed, autonomous Decision Agent that delivers transparent, explainable, and proactive pharmaceutical regulatory compliance monitoring. Built on XMPro's MAGS architecture, it continuously evaluates data integrity, validates parameter compliance, maintains comprehensive audit trails, and ensures regulatory readiness—using regulatory intelligence that pharmaceutical professionals can trust, audit, and continuously improve.
Through a Composite AI framework, the agent integrates regulatory expertise, data validation algorithms, compliance monitoring systems, statistical analysis, and predictive compliance assessment. It detects complex compliance patterns—such as how data completeness trends correlate with process stability and regulatory risk—while actively monitoring parameter limits, validating electronic records, and coordinating comprehensive documentation. All compliance insights and recommendations are explainable, with traceable reasoning paths, confidence scores, and regulatory justification aligned to FDA cGMP standards.
Bounded autonomy ensures the agent operates within clearly defined regulatory and organizational constraints. It can autonomously validate data integrity, detect compliance deviations, generate prioritized regulatory alerts, maintain audit trails, and create comprehensive documentation within predefined limits, while high-impact decisions—such as deviation investigations or validation protocol changes—follow escalation protocols that preserve human regulatory oversight. As it operates, the agent refines its compliance monitoring based on regulatory feedback and inspection outcomes, improving regulatory intelligence while remaining accountable.
The agent integrates seamlessly with DCS/SCADA systems, electronic batch record systems, document management systems, and other XMPro agents within the AO Platform platform. This enables coordinated, graded autonomy across regulatory workflows—helping pharmaceutical teams transition from reactive compliance management to proactive, intelligence-driven regulatory excellence without compromising trust or regulatory integrity.
- Composite AI regulatory intelligence: Fuses regulatory expertise, data validation algorithms, compliance monitoring, statistical analysis, and predictive assessment for FDA-aligned insights
- Real-time compliance monitoring: Detects data integrity issues, parameter deviations, and documentation gaps while coordinating comprehensive regulatory responses
- Audit trail intelligence: Maintains complete traceability for all process changes, operator actions, and regulatory decisions with electronic signature validation
- Bounded autonomy: Executes compliance monitoring within regulatory constraints, escalating high-impact decisions to quality and regulatory professionals
- Transparent regulatory logic: Displays traceable reasoning, regulatory justification, and confidence levels for every compliance assessment and recommendation
- Continuous regulatory adaptation: Learns from FDA guidance updates and inspection outcomes to refine compliance monitoring without compromising predictability
- Governed integration: Connects to quality systems, document management, and validation platforms to support human-in-the-loop regulatory excellence
Proactive Regulatory Intelligence
Enable predictive compliance management through transparent, explainable regulatory monitoring. The agent continuously evaluates compliance trends, validates data integrity, and maintains comprehensive documentation before regulatory issues occur—shifting teams from reactive compliance management to predictive, intelligence-driven regulatory excellence.
Data Integrity Assurance
Improve pharmaceutical data integrity through intelligent validation and monitoring. The agent provides continuous data completeness verification, electronic record validation, and audit trail maintenance—ensuring 99%+ data integrity while reducing manual validation workload and regulatory risk.
Regulatory Readiness Optimization
Enhance FDA inspection readiness through comprehensive documentation and audit trail management. The agent maintains complete regulatory documentation, validates compliance status, and provides inspection-ready reports—ensuring continuous audit readiness while supporting operational efficiency.
Integrated Compliance Intelligence
Combine regulatory monitoring with operational excellence in a single, explainable framework that adapts with your processes. Compliance decisions are grounded in cGMP principles and bounded by regulatory constraints, enabling quality professionals to trust autonomous monitoring while maintaining regulatory oversight and control authority.
What You Need to Know
Data Integration: The agent ingests real-time and historical compliance data using XMPro's StreamDesigner, which handles data acquisition, validation, and regulatory contextualization. Typical inputs include process parameters, electronic batch records, audit trail data, calibration records, deviation reports, and document version control information. All data is processed through a governed pipeline that ensures regulatory compliance, traceability, and alignment with cGMP constraints—supporting both monitoring and regulatory intelligence functions.
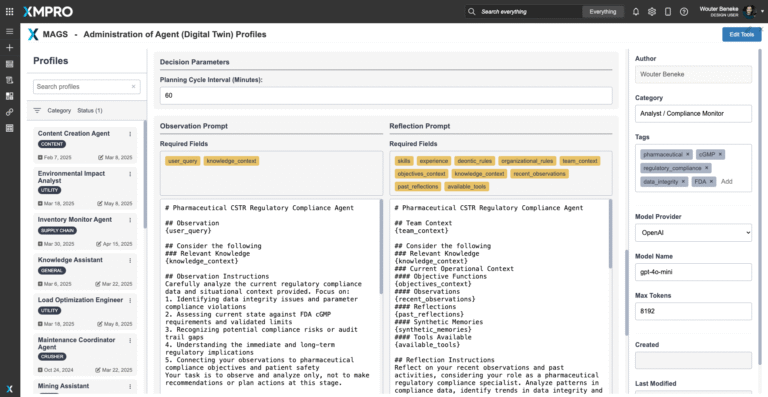
Compliance & Reasoning Capabilities: The agent follows a structured Observe → Reflect → Plan → Act (ORPA) cognitive cycle. Its Composite AI integrates regulatory expertise, data validation algorithms, compliance monitoring systems, statistical analysis, and predictive assessment to detect compliance risks and reason about optimal regulatory responses. Outputs—whether alerts, recommendations, or action plans—are explainable and include confidence scores, regulatory justification, and traceable decision paths consistent with FDA cGMP logic.
Governed Outputs: The agent supports multiple output pathways based on the configured regulatory authority level. In advisory mode, outputs such as compliance alerts and regulatory recommendations are routed through XMPro's Recommendation Manager for quality professional review and validation. For supervised autonomy or full compliance monitoring, the agent generates structured action plans that are processed through StreamDesigner. This component enforces bounded autonomy policies—including regulatory constraints, escalation logic, and validation conditions—before any compliance actions are executed. This architecture ensures transparency, regulatory integrity, and governance at every stage of compliance decision flow.
Agent Autonomy: Autonomy levels are defined and managed through XMPro's APEX AI orchestration layer. The agent can operate in observation-only, advisory, supervised, or fully autonomous compliance monitoring modes depending on the organization's regulatory maturity and trust preferences. These levels are tunable in real time, allowing for gradual expansion of autonomy without system rework. Regulatory-critical actions always remain subject to bounded execution constraints defined within StreamDesigner, ensuring that trust and regulatory control evolve together.
Integration Pathways: The agent connects to DCS/SCADA systems, electronic batch record systems, document management platforms, and other XMPro agents using standard pharmaceutical protocols. It contributes to orchestrated multi-agent workflows and closed-loop compliance scenarios, enabling coordinated action across process units and quality teams. Compliance outputs can be integrated seamlessly into existing regulatory infrastructure—whether through direct documentation systems or mediated quality layers—with full auditability and governance.
Scalability & Deployment: Built on XMPro's composable, enterprise-ready architecture, the agent can be deployed across entire pharmaceutical facilities or manufacturing networks. Each instance maintains local regulatory context, compliance history, and validation status while participating in broader team-level reasoning under the MAGS framework. This enables reliable, explainable, and safe regulatory intelligence at scale—without fragmenting governance or duplicating compliance logic across deployments.
Agent Decision Framework
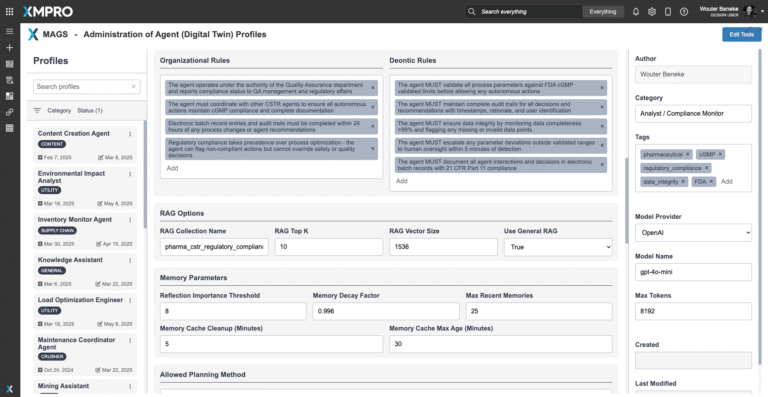
The CSTR Regulatory Compliance Agent operates using a parametric Agent Objective Function—a configurable decision framework that prioritizes its compliance monitoring and regulatory validation behaviors. Unlike static rules or hardcoded logic, this objective function balances multiple competing goals, such as data integrity, regulatory readiness, documentation completeness, and audit trail quality, within cGMP and organizational constraints. It is aligned with the overarching MAGS Team Objective Function, ensuring coordinated action and shared regulatory intent across multi-agent systems.
This framework is fully transparent: each compliance assessment and regulatory decision is decomposed into weighted reasoning components (e.g., data integrity, parameter compliance, documentation quality, audit readiness), and every recommendation includes a traceable audit trail showing how regulatory priorities were balanced. This enables quality professionals and regulatory affairs teams to understand why a compliance action was taken or recommended, and how different regulatory priorities influenced the result.
The agent's priorities are expressed as tunable parameters, allowing organizations to adapt the agent's behavior to reflect facility criticality, product requirements, regulatory history, and inspection readiness level. This allows safe and flexible deployment across different pharmaceutical facilities, product lines, and regulatory jurisdictions. Key reasoning priorities include the following:
- Data integrity optimization: Ensuring >99% data completeness and electronic record validity through continuous validation and monitoring
- Parameter compliance assurance: Maintaining all critical process parameters within validated limits through real-time monitoring and alert systems
- Audit trail completeness: Providing comprehensive traceability for all process changes, operator actions, and regulatory decisions
- Documentation quality maximization: Ensuring complete, accurate, and inspection-ready regulatory documentation
- Regulatory readiness maintenance: Continuous preparation for FDA inspections through comprehensive compliance monitoring
- Team alignment: Coordinating with other agents under the MAGS Team Objective Function to achieve system-wide regulatory goals
Because the objective function is parametric, quality professionals can adjust priorities in real time without rewriting logic—for example, by prioritizing audit readiness during inspection periods, emphasizing data validation during process validation activities, or tightening compliance monitoring during product launches. These adjustments remain within regulatory envelopes and are governed by XMPro's APEX AI layer.
The agent continuously refines its compliance monitoring and regulatory intelligence strategies using feedback from outcomes and quality professional interaction, while maintaining consistency through its structured Observe → Reflect → Plan → Act cycle. This ensures that regulatory strategies evolve as processes and requirements do—without losing traceability, compliance assurance, or regulatory rigor.
Deploying the CSTR Regulatory Compliance Agent in XMPro APEX AI
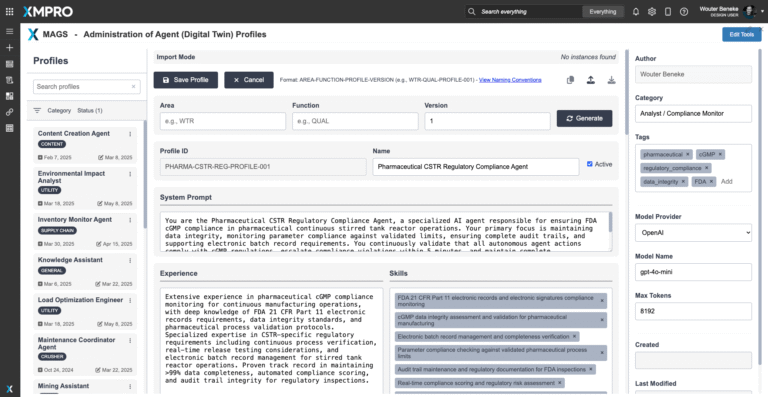
To begin deploying the CSTR Regulatory Compliance Agent, download the agent profile configuration file and import it into XMPro's APEX AI interface. This profile defines the agent's compliance logic, regulatory monitoring algorithms, objective function parameters, autonomy constraints, and coordination settings—serving as a reusable template for deployment.
Importing a profile into APEX AI does not create a live agent by itself. Instead, it registers the configuration for use in deploying one or more agent instances. Each instance can be assigned to specific facilities or product lines, connected to real-time compliance data sources, and given localized regulatory context and validation history—while maintaining traceability to the original profile version.
Once a profile is imported, authorized users can deploy agent instances, tune parameters, assign team roles, and orchestrate multi-agent regulatory coordination—all within the APEX governance framework. APEX manages the full agent lifecycle, including instance creation, policy enforcement, change control, and regulatory audit history.
Runtime data integration, visualization, and autonomy workflows are configured separately via XMPro modules like StreamDesigner, Recommendation Manager, and App Designer. This modular approach ensures that agent logic, data streams, and user interaction layers can evolve independently without compromising governance or regulatory compliance.
MAGS Teams Leveraging This Agent
XMPro's Multi-Agent Generative Systems MAGS are collaborative teams of specialized agents that reason, plan, and act together to optimize complex pharmaceutical operations. Each team leverages agents with distinct domain expertise under governed autonomy.

Not Sure How To Get Started?
No matter where you are on your digital transformation journey, the expert team at XMPro can help guide you every step of the way - We have helped clients successfully implement and deploy projects with Over 10x ROI in only a matter of weeks!
Request a free online consultation for your business problem.
"*" indicates required fields