CSTR Quality Control Agent
Introduction
In pharmaceutical manufacturing, quality failures can cost millions and put patients at risk. Yet most facilities discover quality problems only after batches fail final testing - when it's too late and expensive to recover. The challenge isn't just making product; it's ensuring every single batch meets stringent pharmaceutical specifications before costly downstream processing begins.
Traditional quality control relies on end-of-batch laboratory testing, creating a dangerous blind spot during the critical hours when quality is actually being determined. By the time problems are detected, entire batches may be lost, equipment may need extensive cleaning, and production schedules are disrupted.
The CSTR Quality Control Agent represents a fundamental shift from reactive to predictive quality assurance - an AI-powered quality watchdog that continuously monitors process indicators, detects the earliest signs of quality drift, and prevents deviations before they compromise batch integrity. This agent doesn't optimize for maximum output; it watches vigilantly to ensure every batch stays within pharmaceutical specifications.
The Quality Deviation Detection Challenge
Pharmaceutical manufacturing faces a critical quality detection gap that traditional end-of-batch testing cannot address. Quality deviations often begin hours before they become visible in final product analysis, creating an expensive blind spot where problems develop undetected until it's too late for corrective action.
Quality Blind Spots During Production
- Late deviation detection: Quality problems discovered only after 6-18 hours of batch processing, when correction is impossible
- Hidden quality drift: Gradual parameter changes that slowly push batches toward specification limits go unnoticed
- Process-quality disconnection: Operators focus on process targets while quality indicators drift independently
- Batch consistency variability: Batch-to-batch variations in pH, conductivity, and process conditions create quality uncertainty
- Statistical control gaps: Lack of real-time statistical process control allows quality trends to develop unchecked
Expensive Quality Failures
- Batch losses: Failed batches cost $500K-$2M in lost materials, processing time, and equipment utilization
- Investigation overhead: Quality deviations trigger expensive investigations, documentation, and regulatory reporting
- Schedule disruption: Quality failures disrupt production schedules and delay product delivery commitments
- Cleaning and rework: Equipment cleaning between failed and successful batches consumes significant time and resources
- Regulatory risk: Quality problems create FDA compliance risks and potential inspection findings
Process Control vs. Quality Assurance Conflict
- Optimization pressure: Process control systems push for maximum efficiency while quality requirements demand consistency
- Parameter trade-offs: Operating conditions that maximize yield may compromise quality consistency
- Real-time quality measurement gaps: Critical quality attributes cannot be measured in real-time, requiring surrogate indicators
- Statistical control complexity: Maintaining statistical process control across multiple parameters exceeds human monitoring capacity
- Quality prediction limitations: Correlating real-time process data with final quality outcomes requires advanced statistical modeling
Regulatory Compliance Pressure
- cGMP documentation requirements: Every quality decision must be documented with complete rationale and evidence
- Process validation maintenance: Quality systems must demonstrate continued process control and capability
- Deviation reporting obligations: Quality problems trigger formal deviation procedures and regulatory notifications
- Batch record integrity: Quality decisions must be integrated into electronic batch records with full traceability
- FDA inspection readiness: Quality control systems must demonstrate proactive monitoring and control capabilities
The Cost of Quality Reactivity
These quality detection gaps create a vicious cycle of reactive quality management where problems are addressed only after they impact final product specifications. This reactive approach leads to batch losses, schedule disruptions, investigation overhead, and regulatory compliance risks that could be prevented through proactive quality monitoring and early deviation detection.
Breaking this cycle requires more than additional laboratory testing or enhanced documentation. It demands an intelligent, vigilant quality watchdog that continuously monitors process indicators, detects the earliest signs of quality drift, and prevents deviations before they compromise pharmaceutical specifications.
XMPro CSTR Quality Control Agent
Your 24/7 AI Quality Specialist That Never Misses A Deviation
The CSTR Quality Control Agent is an autonomous pharmaceutical quality guardian that continuously monitors batch consistency, detects quality deviations hours before they appear in final testing, and prevents specification failures through proactive intervention. Unlike process optimization agents that push for maximum output, this agent acts as a vigilant quality watchdog - always watching, always protecting batch integrity.
Operating within XMPro's APEX AI orchestration layer, this agent uses Composite AI to correlate real-time process indicators (pH, conductivity, turbidity) with final product quality, providing early warning when batches begin drifting toward specification limits. It maintains veto power over process changes that could compromise quality, ensuring that pharmaceutical specifications are never sacrificed for production efficiency.
The agent's bounded autonomy focuses exclusively on quality protection - it can halt processes showing quality risk, adjust parameters to maintain consistency, and escalate to human quality personnel when deviations exceed statistical control limits. Every decision includes complete audit trails and statistical evidence, ensuring FDA compliance while preventing costly batch failures.
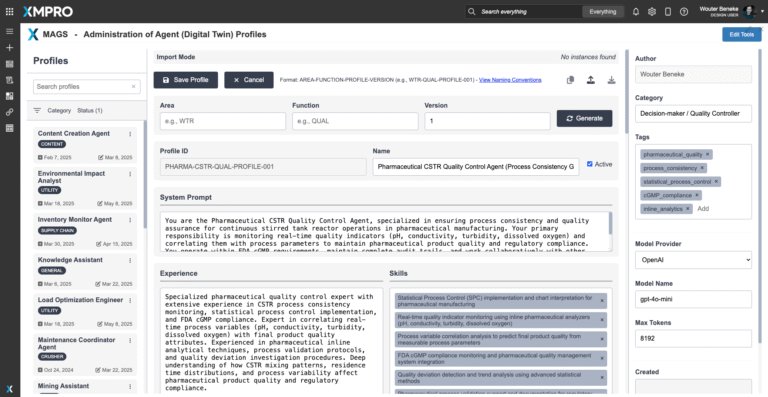
Agent Profile Summary
Meet Your New Quality Deviation Watchdog
The XMPro CSTR Quality Control Agent is an autonomous quality guardian designed specifically to detect, prevent, and resolve quality deviations before they compromise pharmaceutical batch specifications. Running within the APEX AI orchestration layer, it continuously monitors quality indicators, maintains statistical process control, and acts as the final quality authority within multi-agent teams.
Unlike process optimization agents that focus on maximizing output, this agent prioritizes batch consistency and specification compliance above all else. It uses advanced statistical process control, real-time quality correlation analysis, and predictive quality modeling to detect the earliest signs of quality drift - often 2-4 hours before problems become visible in traditional testing.
All quality decisions are explainable and include statistical evidence, confidence intervals, and complete audit trails for FDA compliance. The agent maintains veto authority over process changes that could impact quality, ensuring that pharmaceutical specifications are never compromised for production efficiency. When quality risks are detected, it can automatically adjust parameters, halt processes, or escalate to human quality personnel based on configured thresholds.
Fully integrated with electronic batch records, laboratory systems, and quality management systems, the CSTR Quality Control Agent serves as an intelligent quality watchdog - helping organizations shift from reactive quality management to proactive deviation prevention and batch protection.
- Quality deviation detection: Identifies quality drift 2-4 hours before traditional testing through statistical correlation analysis
- Batch consistency monitoring: Maintains process capability and reduces batch-to-batch variation through real-time statistical control
- Veto authority: Can override process optimization decisions that pose quality risks, with complete audit trail documentation
- FDA compliance integration: Automated documentation and audit trails meeting 21 CFR Part 11 requirements
- Predictive quality modeling: Correlates real-time process indicators with final product quality for early intervention
- Statistical process control: Implements advanced SPC methods and control charting for pharmaceutical manufacturing
Quality Risk Reduction and Batch Protection
Prevent costly batch failures through early deviation detection and proactive quality intervention. Reduce batch loss rates from 2-5% to less than 0.5% through predictive quality monitoring and statistical process control.
Cost Avoidance and Waste Reduction
Avoid $500K-$2M batch losses by detecting quality problems while corrective action is still possible. Reduce investigation costs, cleaning overhead, and schedule disruptions through proactive quality management.
Regulatory Compliance Excellence
Maintain FDA compliance through automated documentation, statistical validation, and complete audit trails. Demonstrate proactive quality control capabilities during regulatory inspections and audits.
Process Consistency and Capability Improvement
Improve process capability indices (Cpk) from 1.0-1.2 to 1.5-2.0 through advanced statistical process control and real-time quality correlation analysis. Reduce batch-to-batch variation and enhance product consistency.
What You Need to Know
Quality Data Integration: Ingests real-time quality indicators through XMPro's StreamDesigner. Typical inputs include pH (±0.1 unit precision), conductivity (±2% accuracy), turbidity (<10 NTU), dissolved oxygen, temperature-corrected measurements, and historical batch quality data.
Statistical Analysis Capabilities: Operates through continuous statistical process control including X-bar and R charts, process capability analysis, correlation modeling, trend detection, and predictive quality assessment. Uses advanced statistical methods including multivariate analysis and quality prediction algorithms.
Quality Authority Outputs: Provides quality assessments, batch consistency evaluations, deviation alerts, and process constraint recommendations through XMPro's Recommendation Manager. All outputs include statistical evidence, confidence levels, and complete audit trail documentation.
Agent Autonomy: Operates with quality-focused bounded autonomy configured in XMPro's APEX AI orchestration layer. Can autonomously adjust quality-related parameters, halt processes showing quality risk, and escalate deviations exceeding statistical control limits.
Integration Pathways: Connects with laboratory information systems (LIMS), electronic batch records, quality management systems, and process control systems. Supports closed-loop quality control workflows and multi-agent coordination with veto authority.
Scalability & Deployment: Designed for pharmaceutical manufacturing environments with multiple agents monitoring batch quality across production lines. Each agent maintains batch-specific quality models while contributing to facility-wide quality intelligence.
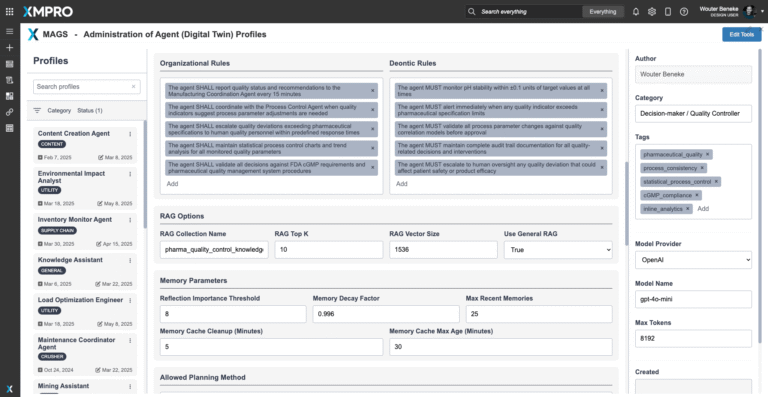
Quality Protection Decision Framework
The CSTR Quality Control Agent operates with an internal Quality Protection Objective Function that prioritizes batch specification compliance and consistency above all other considerations. This objective function is specifically designed to act as a quality watchdog, with authority to constrain or override other operational decisions when quality risks are detected.
The agent's decision framework balances multiple quality priorities while maintaining its primary mission of preventing specification failures:
- Specification compliance: Ensuring all quality indicators remain within pharmaceutical specifications with appropriate statistical confidence
- Process consistency: Maintaining batch-to-batch consistency through statistical process control and variation reduction
- Early deviation detection: Maximizing lead time for quality intervention by detecting trends before they become critical
- Statistical confidence: Ensuring all quality decisions are supported by statistically valid evidence and appropriate confidence levels
- Regulatory compliance: Maintaining complete audit trails and documentation meeting FDA cGMP requirements
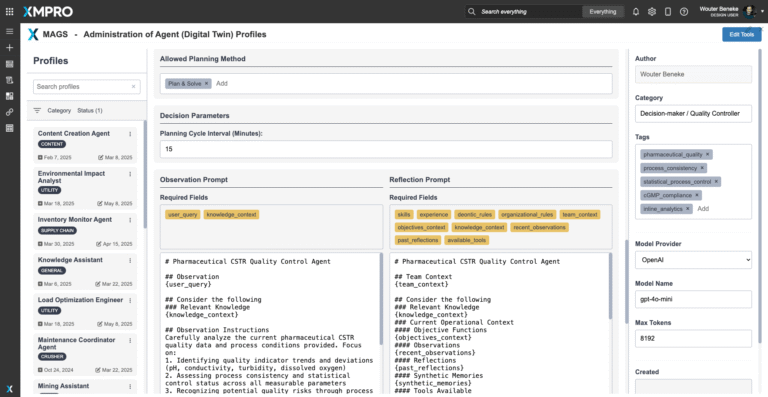
The agent's quality-focused objective function enables dynamic decision-making based on current batch conditions and quality risk assessment. For example:
- High-risk quality situations trigger conservative quality protection with tighter control limits
- Stable quality conditions allow normal statistical control with standard specification limits
- Novel quality patterns trigger enhanced monitoring and human quality expert consultation
- Critical quality deviations override all other operational priorities and require immediate intervention
The agent continuously refines its quality models through statistical analysis of batch outcomes and quality performance. This learning capability ensures that quality protection strategies improve over time while maintaining pharmaceutical manufacturing standards.
Importing and Deploying the Quality Control Agent in XMPro APEX AI
To deploy the CSTR Quality Control Agent, download the agent profile JSON configuration file and access the XMPro APEX AI interface. APEX AI provides governance and lifecycle management for Decision Agents across XMPro's AO Platform.
Import the agent profile through APEX AI, which includes the agent's quality protection parameters, statistical control limits, bounded autonomy settings, and FDA compliance requirements. After import, use XMPro's StreamDesigner to configure real-time data connections to your laboratory systems, inline analyzers, electronic batch records, and quality management systems.
Once deployed, the agent operates within the defined quality governance framework and statistical control limits. It begins continuous quality monitoring immediately, learning from batch patterns and contributing quality protection decisions to pharmaceutical manufacturing workflows. Ongoing quality parameter tuning and statistical model adjustments can be performed through APEX AI to ensure alignment with evolving quality requirements and regulatory standards.
MAGS Teams Leveraging This Agent
XMPro's Multi-Agent Generative Systems MAGS are collaborative teams of specialized agents that reason, plan, and act together to optimize complex pharmaceutical operations. Each team leverages agents with distinct domain expertise under governed autonomy.
How XMPro AO Platform Modules Enable the Quality Control Agent

Not Sure How To Get Started?
No matter where you are on your digital transformation journey, the expert team at XMPro can help guide you every step of the way - We have helped clients successfully implement and deploy projects with Over 10x ROI in only a matter of weeks!
Request a free online consultation for your business problem.
"*" indicates required fields